Chapter 11: Q 11-11-41 E-b (page 350)
Which compound in each of the following pairs will react faster in an SN2reaction with OH-?
(b) CH3CH2Iin ethanol or dimethyl sulfoxide
Short Answer
CH3CH2I
Learning Materials
Features
Discover
Chapter 11: Q 11-11-41 E-b (page 350)
Which compound in each of the following pairs will react faster in an SN2reaction with OH-?
(b) CH3CH2Iin ethanol or dimethyl sulfoxide
CH3CH2I
All the tools & learning materials you need for study success - in one app.
Get started for free
SN2 reactions take place with inversion of configuration, andSN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of configuration. Propose a mechanism.
.
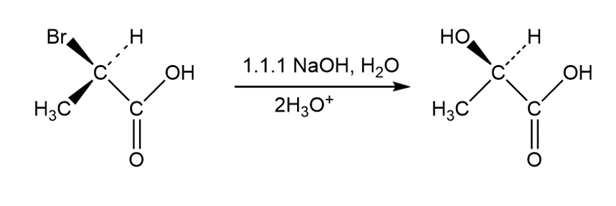
Question:What products would you expect from the reaction of 1-bromopropane with each of the following?
(d) NaCN
Question: Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane:
(b)
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
a)
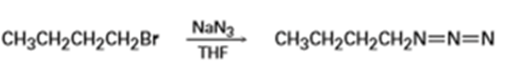
What product would you expect to obtain from reaction of with (R)-2-bromobutane? Show the stereochemistry of both the reactant and product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.