Chapter 11: 45bE (page 350)
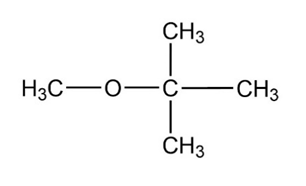
How might you prepare each of the following molecules using a nucleophilic substitution reaction at some step?
Learning Materials
Features
Discover
Chapter 11: 45bE (page 350)
How might you prepare each of the following molecules using a nucleophilic substitution reaction at some step?

All the tools & learning materials you need for study success - in one app.
Get started for free
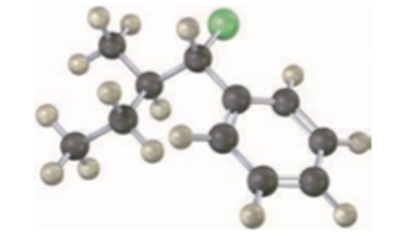
Draw the structure and assign Z or E stereochemistry to the product you expect from E2 reaction of the following molecule with NaOH (green = Cl):
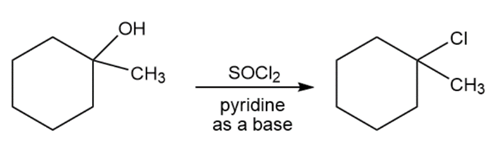
Question: The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actual product.
c.
Which reaction in each of the following pairs would you expect to be faster?
(b) Thedisplacement byon bromoethane or on bromocyclohexane
Write the product you would expect from reaction of each of the following alkyl halides with (1)Na+-SCH3and (2)Na+-OH (green = Cl):
c)
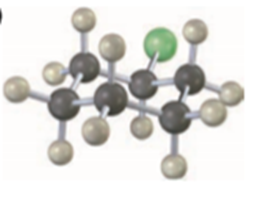
Question: Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane:
(c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.