Chapter 11: 45aE (page 350)
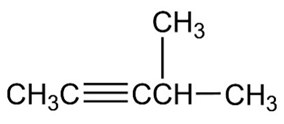
How might you prepare each of the following molecules using a nucleophilic substitution reaction at some step?
(a)
Learning Materials
Features
Discover
Chapter 11: 45aE (page 350)
How might you prepare each of the following molecules using a nucleophilic substitution reaction at some step?
(a)

All the tools & learning materials you need for study success - in one app.
Get started for free
d) Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
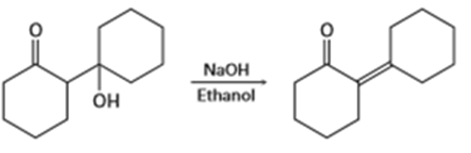
Question: What stereochemistry do you expect for the alkene obtained by E2 elimination of (1R, 2R)-1,2-dibromo-1,2-diphenylethane? Draw a Newman projection of the reacting conformation.
Which compound in each of the following pairs will react faster in an SN2reaction with OH-?
(b) CH3CH2Iin ethanol or dimethyl sulfoxide
Methyl esters () undergo a cleavage reaction to yield carboxylate ions plus iodomethane on heating with LiI in dimethylformamide:
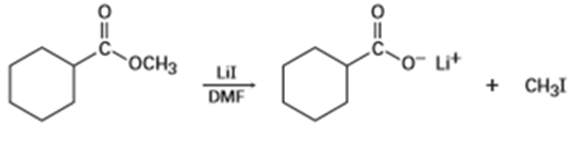
The following evidence has been obtained: (1) The reaction occurs much faster in DMF than in ethanol. (2) The corresponding ethyl ester () cleaves approximately 10 times more slowly than the methyl ester. Propose a mechanism for the reaction. What other kinds of experimental evidence could you gather to support your hypothesis?
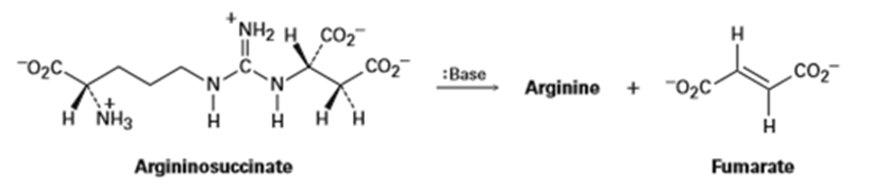
One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.
What do you think about this solution?
We value your feedback to improve our textbook solutions.