Chapter 3: Q47E (page 88)
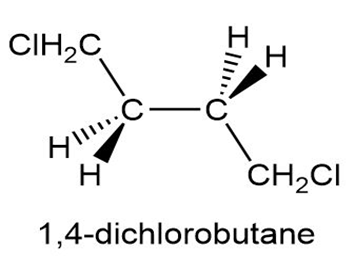
Draw the most stable conformation of 1,4-dichlorobutane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper respectively.
Short Answer
Learning Materials
Features
Discover
Chapter 3: Q47E (page 88)
Draw the most stable conformation of 1,4-dichlorobutane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper respectively.

All the tools & learning materials you need for study success - in one app.
Get started for free
Construct a qualitative potential energy diagram for rotation about the C-C bond of 1,2-dibromo ethane. Which conformation would you expect to be most stable? Label the anti and gauche conformations of 1,2-dibromo ethane.
Draw the structures of the following molecules:
(b) Ethylenimine, , a substance used in the synthesis of
melamine polymers; it contains no multiple bonds.
Identify the hydrogen atoms on the compounds shown in Problem 3-8 as primary, secondary, or tertiary.
Sight along the C2-C1 bond of 2-methylpropane (isobutane).
(a) Draw a Newman projection of the most stable conformation.
Sight along the C2-C1 bond of 2-methylpropane (isobutane).
(b) Draw a Newman projection of the least stable conformation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.