Chapter 3: Q46E (page 88)
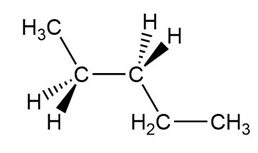
Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.
Short Answer
Learning Materials
Features
Discover
Chapter 3: Q46E (page 88)
Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.

All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the hybridization of the carbon atom in each of the following Functional groups:
(c) Carboxylic acid
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, or quaternary:
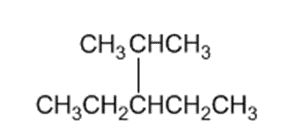
b)
Draw the structures of the following molecules:
(a) Biacetyl, , a substance with the aroma of butter; it containsno rings or carbon–carbon multiple bonds.
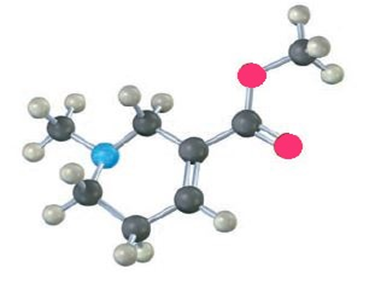
Identify the functional groups in the following model of arecoline, a veterinary drug used to control worms in animals. Convert the drawing into a line-bond structure and a molecular formula (red5O, blue5N).
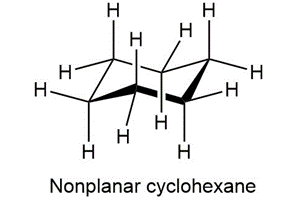
In the next chapter we’ll look at cycloalkanes—saturated cyclic hydrocarbons—and we’ll see that the molecules generally adopt puckered, nonplanar conformations. Cyclohexane, for instance, has a puckered shape like a lounge chair rather than a flat shape. Why?
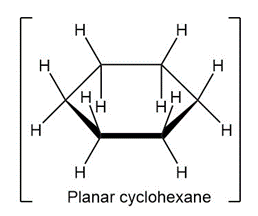
What do you think about this solution?
We value your feedback to improve our textbook solutions.