Chapter 3: Q. 3-3-26E-b (page 88)
Draw the structures of the following molecules:
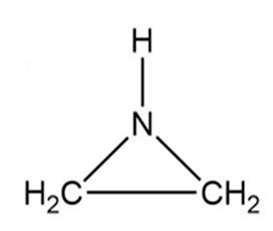
(b) Ethylenimine, , a substance used in the synthesis of
melamine polymers; it contains no multiple bonds.
Short Answer
(b)
Learning Materials
Features
Discover
Chapter 3: Q. 3-3-26E-b (page 88)
Draw the structures of the following molecules:
(b) Ethylenimine, , a substance used in the synthesis of
melamine polymers; it contains no multiple bonds.
(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Name the eight 5-carbon alkyl groups you drew in Problem 3-7.
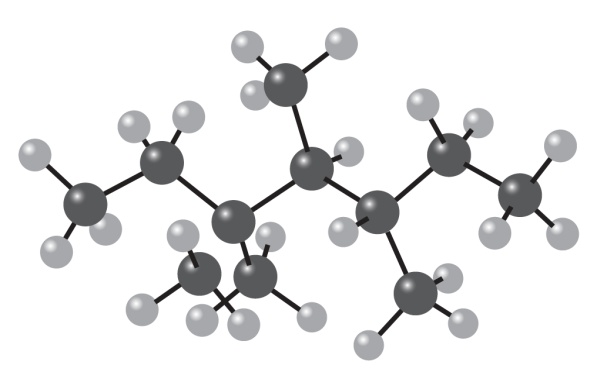
Question: Give the IUPAC name for the following hydrocarbon, and convert the drawing into a skeletal structure.
Question: Sight along the C2-C3 bond of 2,3-dimethylbutane, and draw a Newman projection of the most stable conformation.
Give IUPAC names for the following alkanes, and convert each drawing
into a skeletal structure.
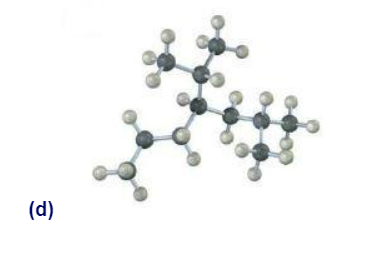
Sight along the C2-C1 bond of 2-methylpropane (isobutane).
(d) Assign relative values to the maxima and minima in your graph, given that
An eclipsing interaction costs 4.0 kJ/mol and an eclipsing
Interaction costs 6.0 kJ/mol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.