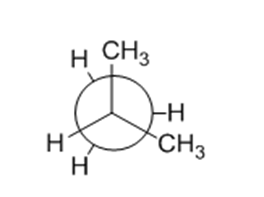
Chapter 3: Q. 3-3-16 P-b (page 85)
Sight along the C2-C1 bond of 2-methylpropane (isobutane).
(b) Draw a Newman projection of the least stable conformation.
Short Answer
(b)
Learning Materials
Features
Discover
Chapter 3: Q. 3-3-16 P-b (page 85)
Sight along the C2-C1 bond of 2-methylpropane (isobutane).
(b) Draw a Newman projection of the least stable conformation.
(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
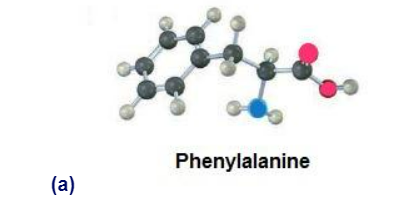
Identify the functional groups in the following substances, and convert
each drawing into a molecular formula (red 5 O, blue 5 N).
Construct a qualitative potential energy diagram for rotation about the C-C bond of 1,2-dibromo ethane. Which conformation would you expect to be most stable? Label the anti and gauche conformations of 1,2-dibromo ethane.
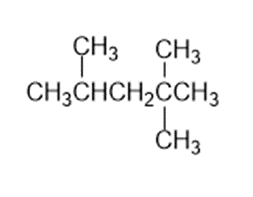
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, or quaternary:
C)
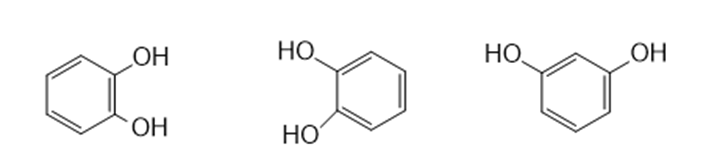
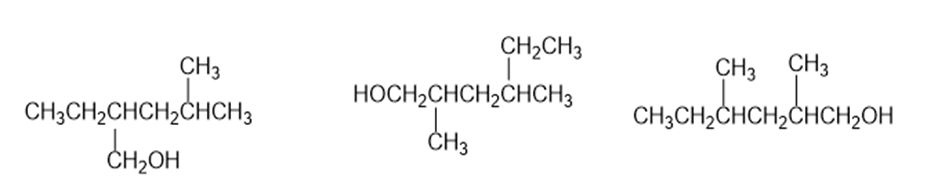
In each of the following sets, which structures represent the same compoundand which represent different compounds?
(b)
In each of the following sets, which structures represent the same compoundand which represent different compounds?
(c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.