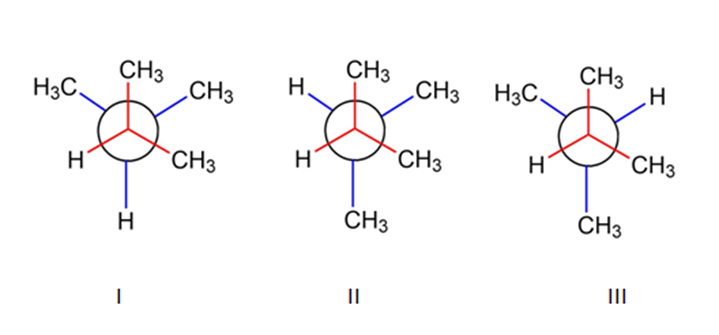
Chapter 3: Q43E (page 88)
What are the relative energies of the three possible staggered conformation around the C-2, C-3 bond in 2,3-dimethyl butane?
Short Answer
Learning Materials
Features
Discover
Chapter 3: Q43E (page 88)
What are the relative energies of the three possible staggered conformation around the C-2, C-3 bond in 2,3-dimethyl butane?

All the tools & learning materials you need for study success - in one app.
Get started for free
Construct a qualitative potential energy diagram for rotation about the C-C bond of 1,2-dibromo ethane. Which conformation would you expect to be most stable? Label the anti and gauche conformations of 1,2-dibromo ethane.
Draw the structures of the following molecules:
(b) Ethylenimine, , a substance used in the synthesis of
melamine polymers; it contains no multiple bonds.
Draw structures of alkanes that meet the following descriptions:
(a) An alkane with two tertiary carbons
Give IUPAC names for the following compounds:
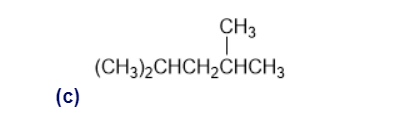
Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.
What do you think about this solution?
We value your feedback to improve our textbook solutions.