Chapter 18: Q66E (page 594)
Identify the reagents a-e in the following scheme:
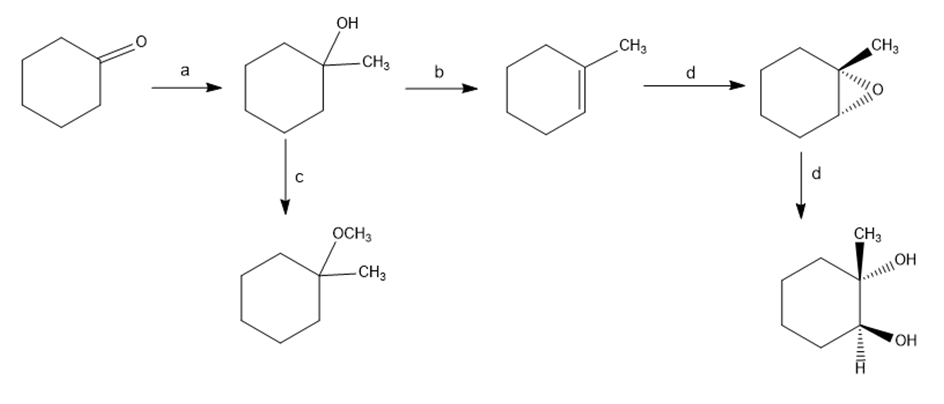
Short Answer
The answer is
Learning Materials
Features
Discover
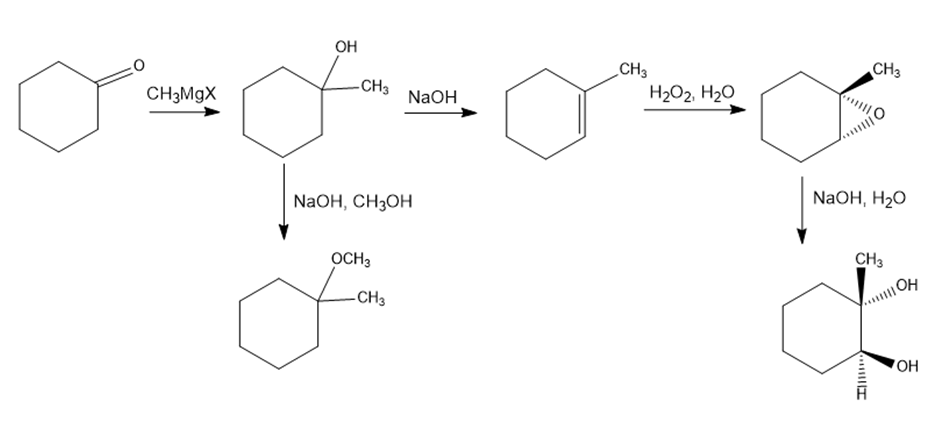
Chapter 18: Q66E (page 594)
Identify the reagents a-e in the following scheme:

The answer is

All the tools & learning materials you need for study success - in one app.
Get started for free
In nature, the enzyme chorismate mutase catalyses a Claisen rearrangement of chorismate that involves both the terminal double bond and the double bond with the highlighted carbon. What is the structure of prephenate, the biological precursor to the amino acids phenylalanine and tyrosine?
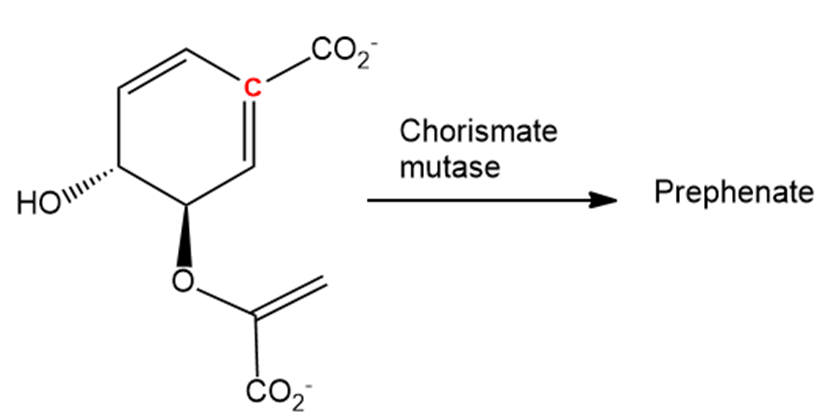
Chorismate
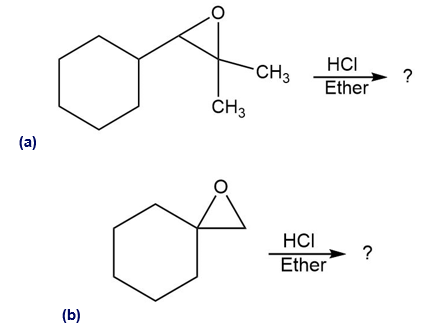
Predict the major product of each of the following reactions:
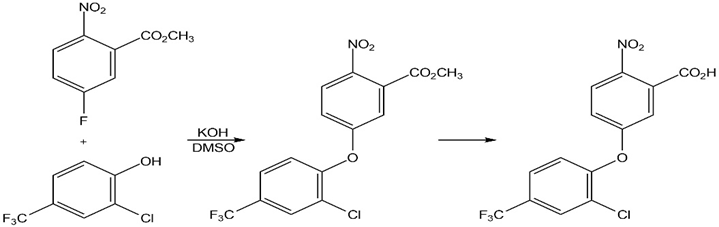
The herbicide acifluorfen can be prepared by a route that begins withreaction between a phenol and an aryl fluoride. Propose a mechanism.
15-Crown-5 and 12-crown-4 ethers complex Na+ and Li+, respectively. Make models of these crown ethers, and compare the sizes of the cavities.
Show the product, including stereochemistry, that would result from reaction of the following epoxide with HBr
What do you think about this solution?
We value your feedback to improve our textbook solutions.