Chapter 18: Q35E (page 594)
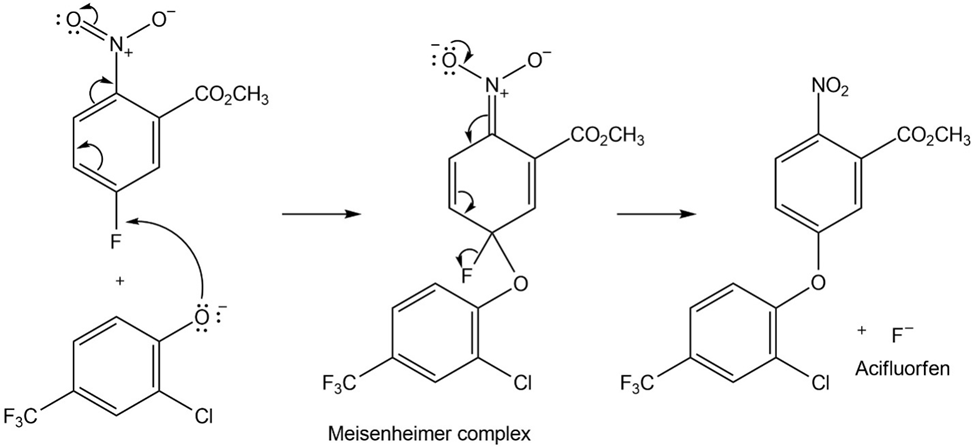
The herbicide acifluorfen can be prepared by a route that begins withreaction between a phenol and an aryl fluoride. Propose a mechanism.
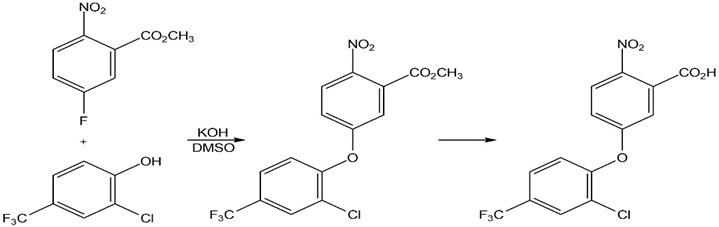
Short Answer
The mechanism of the given reaction is:
Learning Materials
Features
Discover
Chapter 18: Q35E (page 594)
The herbicide acifluorfen can be prepared by a route that begins withreaction between a phenol and an aryl fluoride. Propose a mechanism.

The mechanism of the given reaction is:

All the tools & learning materials you need for study success - in one app.
Get started for free
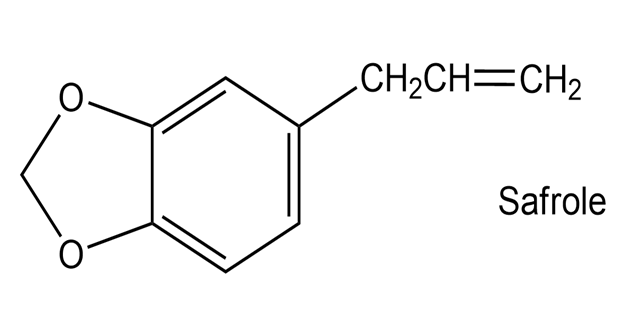
Safrole, a substance isolated from the oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol).
How would you prepare the following diols?
How could you prepare benzyl phenyl ether from benzene and phenol? More than one step is required.
Predict the major product of the following reactions:
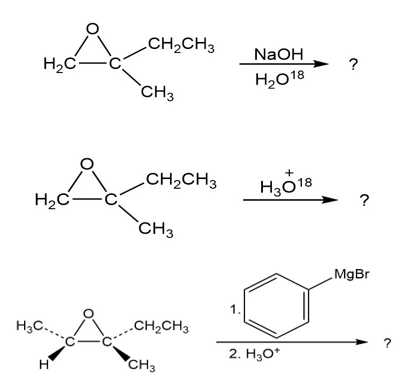
In the formation of the prepolymer used to make epoxy resins, a bisphenol reacts with epichlorohydrin in the presence of a base. Show the product and mechanism when two moles of phenol react with epichlorohydrin.
What do you think about this solution?
We value your feedback to improve our textbook solutions.