Chapter 18: Q12P (page 581)
Predict the major product of each of the following reactions:
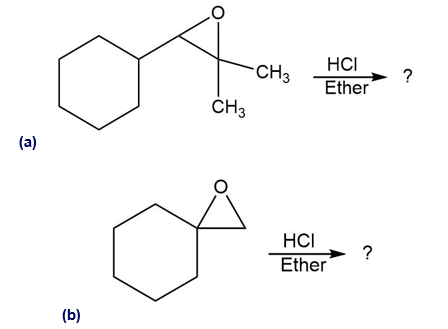
Short Answer
Answer
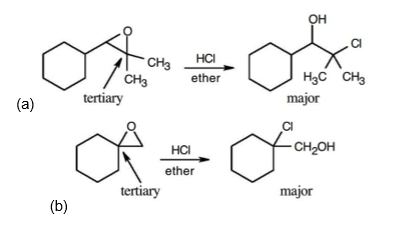
Learning Materials
Features
Discover
Chapter 18: Q12P (page 581)
Predict the major product of each of the following reactions:

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.
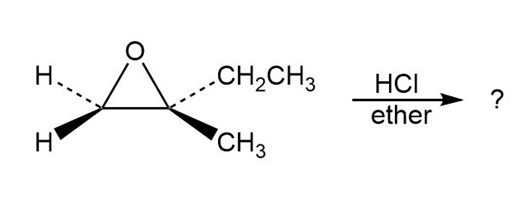
b.
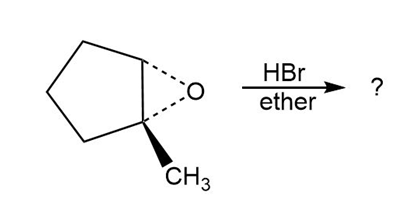
c.
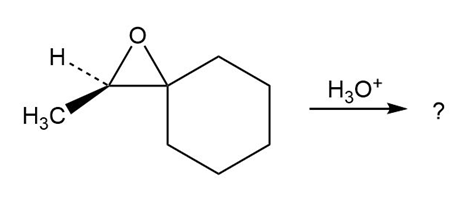
d.
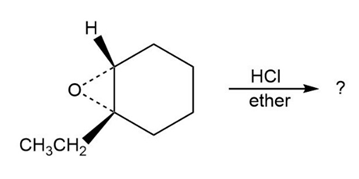
Rank the following halides in order of their reactivity in Williamson synthesis:
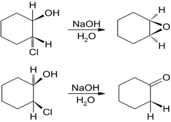
Treatment of trans-2-chlorocyclohexanol with NaOH yields 1,2-epoxycyclohexane,but reaction of the cis isomer under the same conditionsyields cyclohexanone. Propose mechanisms for both reactions, andexplain why the different results are obtained.
We saw in section 17-4 that ketones react with to yield alcohols. We’ll also see in section 22-3 that ketones react with to yield -bromo ketones. Perhaps surprisingly, treatment with of the -bromo ketone from acetophenone yields an epoxide rather than a bromo alcohol. Show the structure of the epoxide, and explain its formation.
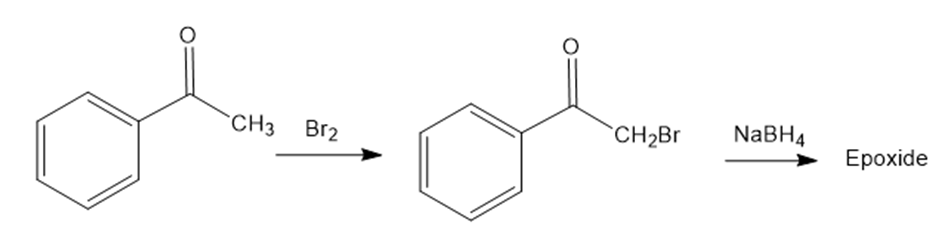
Acetophenone An -bromo ketone
How could you prepare benzyl phenyl ether from benzene and phenol? More than one step is required.
What do you think about this solution?
We value your feedback to improve our textbook solutions.