Step 1:
The step involves the synthesis of sodium phenoxide from phenol. The phenol reacts with a strong base like sodium hydride to eliminate hydrogen molecules. The phenoxide ion and sodium ion form the sodium phenoxide.
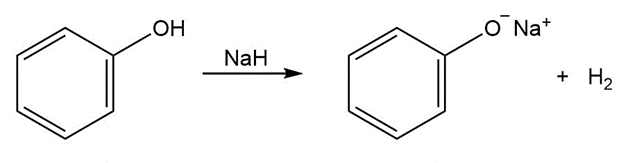
Synthesis of methoxy benzene
Step 2:
The benzene molecule undergoes Friedel-Crafts alkylation to form toluene. It involves the reaction of benzene with methyl chloride in the presence of Lewis acids like aluminium chloride to carry out substitution. The toluene formed reacts with N-bromosuccinimide in the presence of benzoyl peroxide to form benzyl chloride.
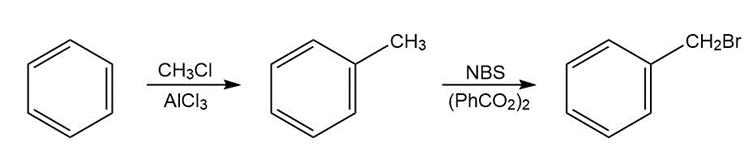
Synthesis of benzyl chloride
Step 3:
The sodium phenoxide formed from phenol and the benzyl chloride formed from benzene undergoes a condensation reaction eliminating sodium bromide to form benzyl phenyl ether.
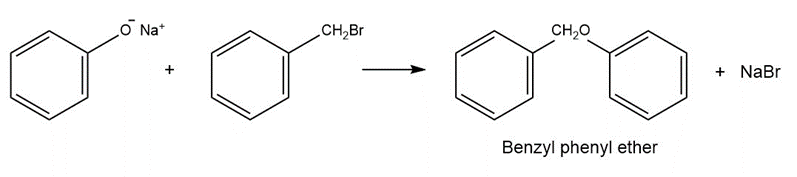
Synthesis of benzyl phenyl ether











