Chapter 18: Q18-18-57 (page 594)
How would you synthesize anethole (Problem 18-54) from phenol?
Short Answer
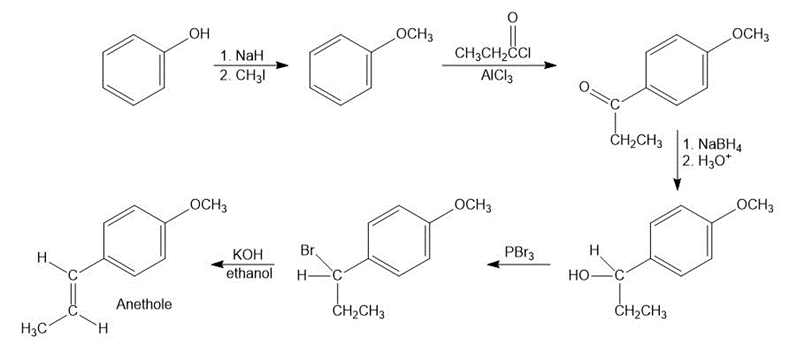
Synthesis of anethole from phenol
Learning Materials
Features
Discover
Chapter 18: Q18-18-57 (page 594)
How would you synthesize anethole (Problem 18-54) from phenol?

Synthesis of anethole from phenol
All the tools & learning materials you need for study success - in one app.
Get started for free
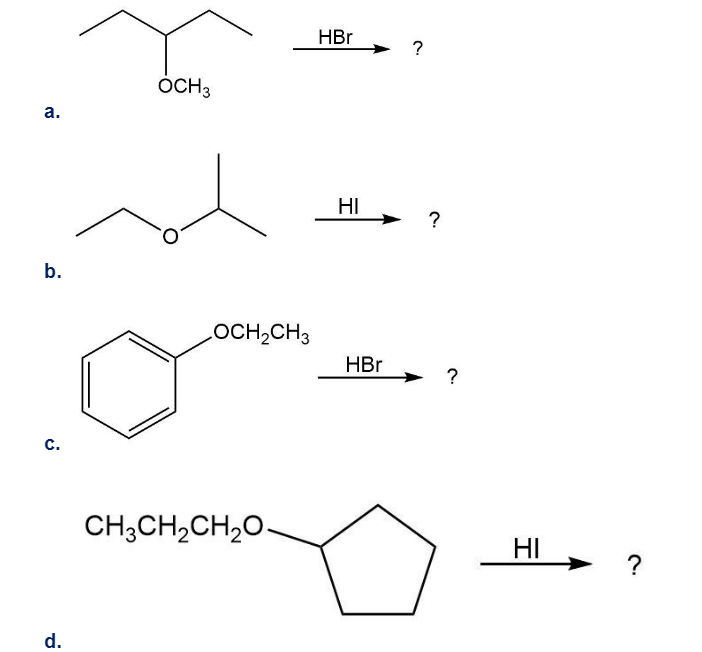
Predict the major product of each of the following reactions:
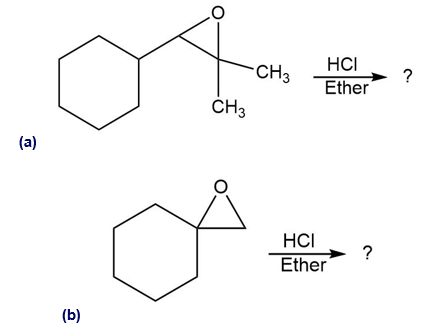
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
Reaction of cis-2-butene with m-chloroperoxybenzoic acid yields an epoxide different from that obtained by reaction of the trans isomer. Explain.
What is the stereochemistry of the product from acid-catalyzed hydrolysis of trans-5,6-epoxydecane? How does the product differ from that formed in Problem 18-47?
How would you prepare the following ethers using a Williamson synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.