Chapter 18: Q28E (page 594)
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.
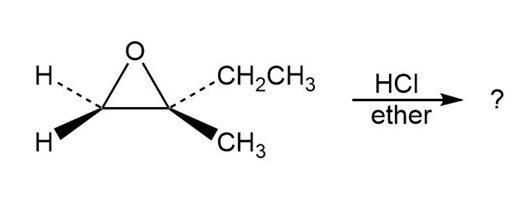
b.
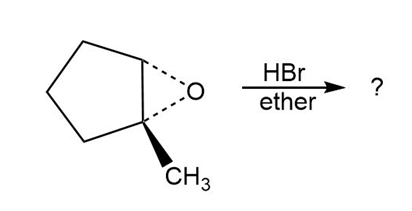
c.
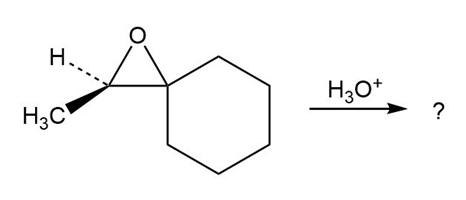
d.
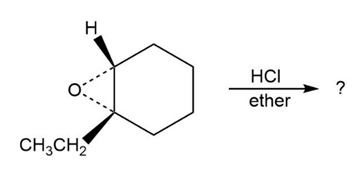
Short Answer
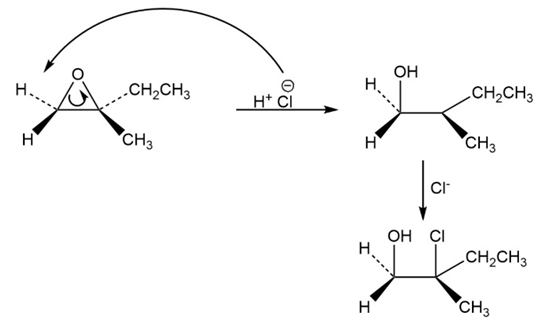
a.
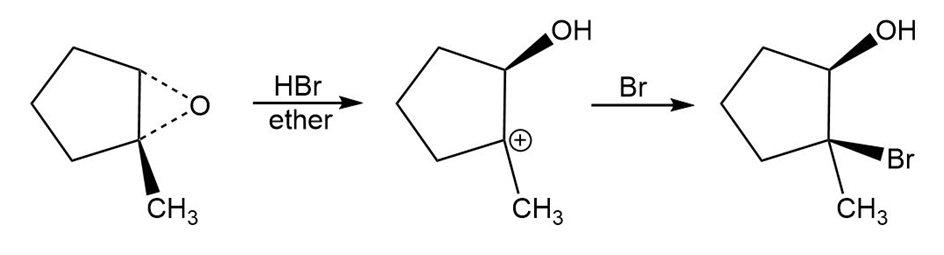
b.
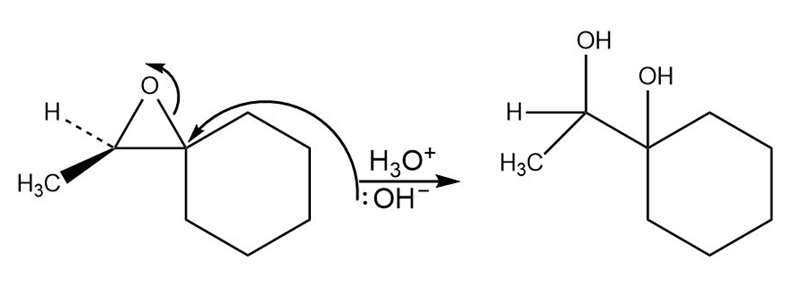
c.
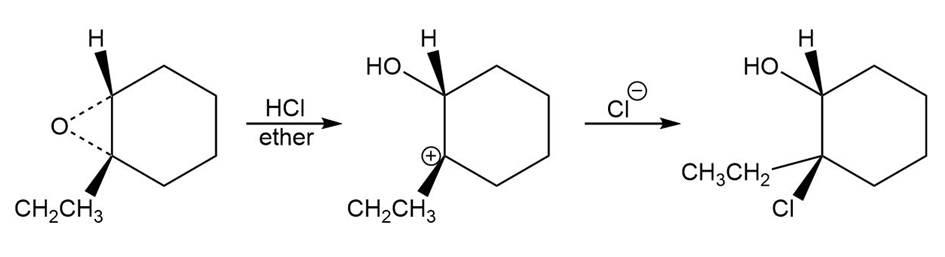
d.
Learning Materials
Features
Discover
Chapter 18: Q28E (page 594)
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.

b.

c.

d.

a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Griginard reagents react with oxetane, a four membered cyclic ether, to yield primary alcohols, but the reaction is much slower than the corresponding reaction with ethylene oxide. Suggest a reason for the difference in reactivity between oxetane and ethylene oxide.
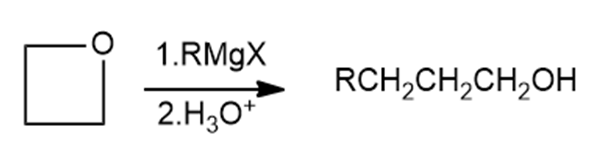
Oxetane
Name the following ethers:
a)
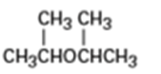
How could you prepare benzyl phenyl ether from benzene and phenol? More than one step is required.
How would you prepare the following ethers using a Williamson synthesis?
Acid-catalyzed hydrolysis of a 1,2-epoxycyclohexane produces a transdiaxial 1,2-diol. What product would you expect to obtain from acidic hydrolysis of cis-3-tert-butyl-1,2-epoxycyclohexane? (Recall that the bulky tert-butyl group locks the cyclohexane ring into a specific conformation.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.