Chapter 18: Q28-E (page 594)
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.
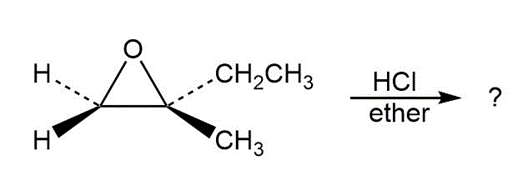
b.
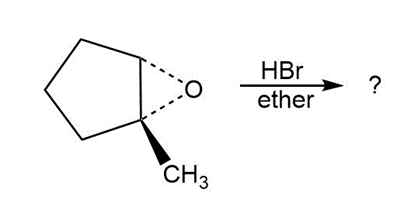
c.
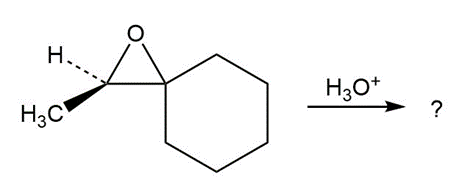
d.
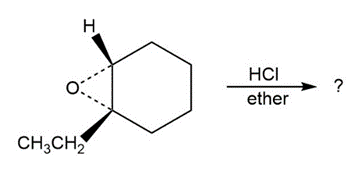
Short Answer
a.
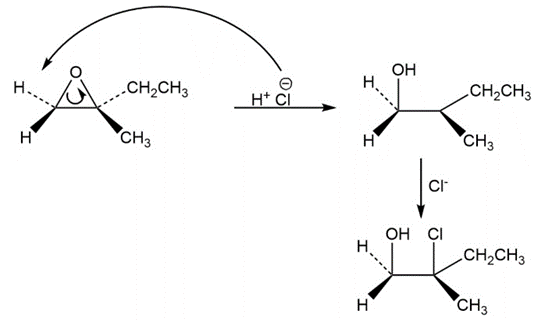
b.
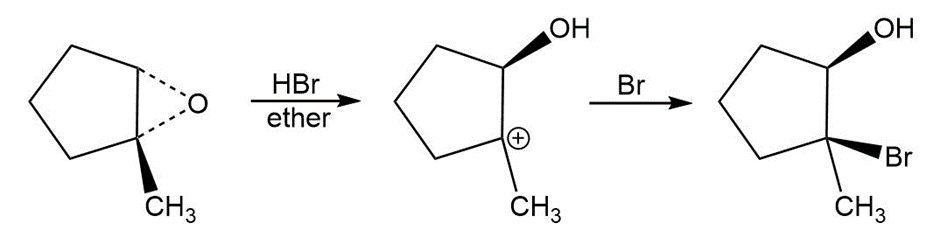
c,
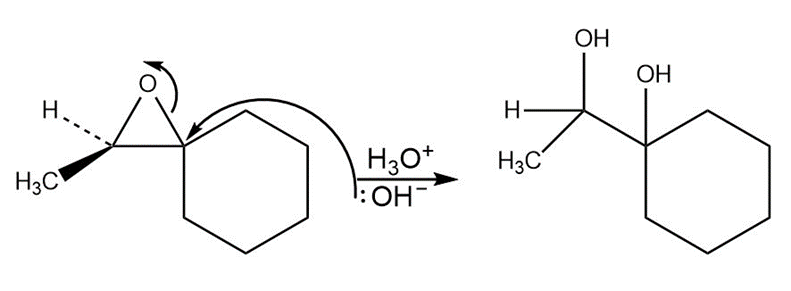
d.
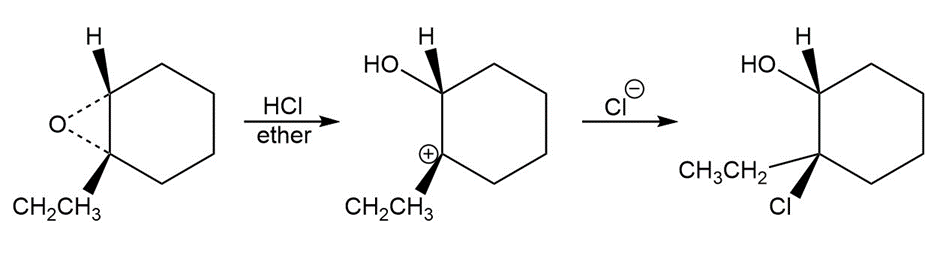
Learning Materials
Features
Discover
Chapter 18: Q28-E (page 594)
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.

b.

c.

d.

a.

b.

c,

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
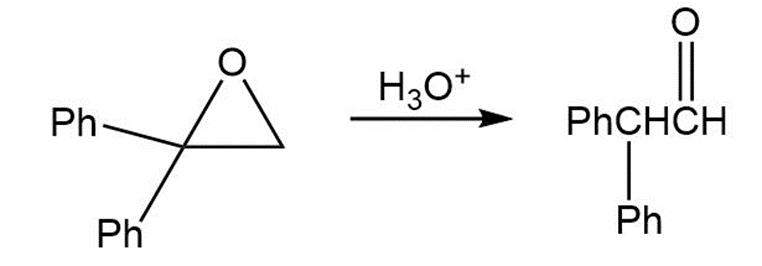
Treatment of 1,1-diphenyl-1,2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction.
Meerwein’s reagent, triethyloxoniumtetrafluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein’s reagent with cyclohexanol, and account for the fact that trialkyloxonium salts are much more reactive alkylating agents than alkyl iodides.
Meerwein’s reagent
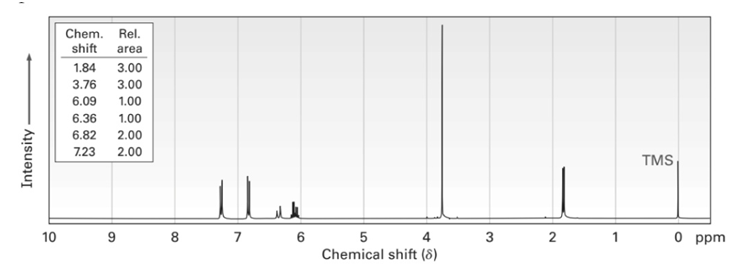
Anethole, , a major constituent of the oil of anise, has the NMR spectrum shown. On oxidation with , anethole yields p-methoxybenzoic acid. What is the structure of anethole? Assign all peaks in the NMR spectrum, and account for the observed splitting patterns
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.
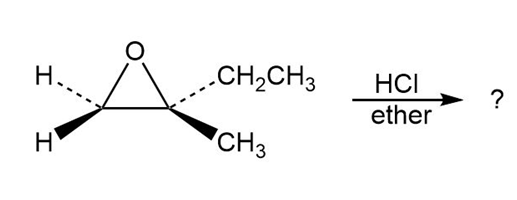
b.
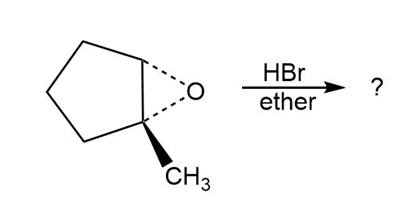
c.
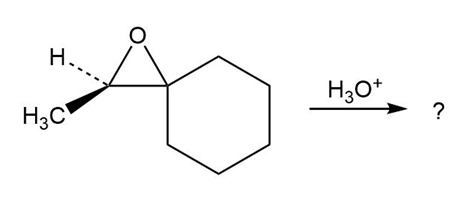
d.
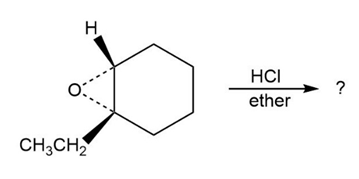
Give IUPAC names for the following structures:
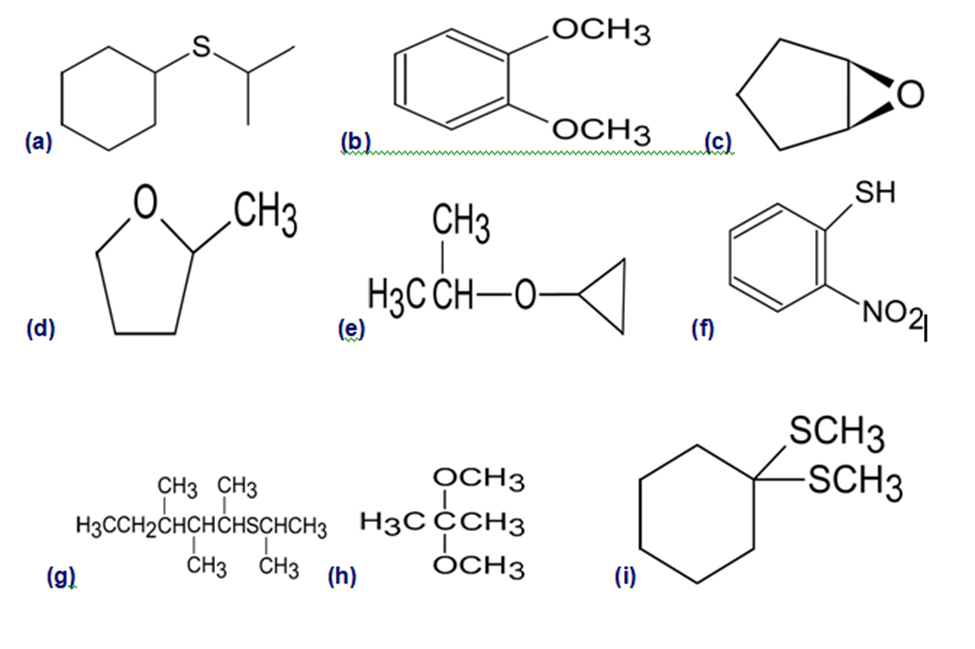
What do you think about this solution?
We value your feedback to improve our textbook solutions.