Chapter 20: Q14P (page 672)
How would you prepare 1-phenyl-2-butanone, , from
benzyl bromide,? More than one step is required.
Short Answer
The preparation of 1-phenyl-2-butanone from benzyl bromide is shown in two step as
follows:
Learning Materials
Features
Discover
Chapter 20: Q14P (page 672)
How would you prepare 1-phenyl-2-butanone, , from
benzyl bromide,? More than one step is required.
The preparation of 1-phenyl-2-butanone from benzyl bromide is shown in two step as
follows:
All the tools & learning materials you need for study success - in one app.
Get started for free
Rank the following compounds in order of increasing acidity. Don’t look at a table of data to help with your answer.
(a) Benzoic acid, p-methylbenzoic acid, p-chlorobenzoic acid
(b) p-Nitrobenzoic acid, acetic acid, benzoic acid
Draw structures corresponding to the following IUPAC names:
Hepatanedioic acid
2-Hexen-4-ynoic acid
4-Ethyl-2-propyloctanoic acid
3-chlorophthalic acid
Triphenylacetic acid
2-Cyclobutenecarbonitrile
m-Benzoyl benzonitrile
3-Methyl-2-hexenoic acid (mixture of E and Z isomers) has been identified as the substance responsible for the odour of human sweat. Synthesize the compound from starting materials having five or fewer carbons
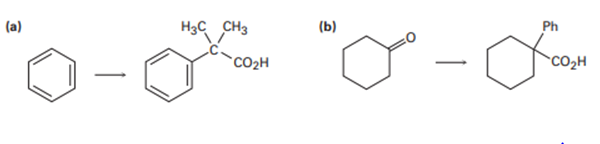
How would you carry out the following transformations? More than one step is needed in each case.
Predict the product (s) and provide the mechanism for each reaction below.
What do you think about this solution?
We value your feedback to improve our textbook solutions.