Chapter 20: Q24E (page 678)
Predict the product (s) and provide the mechanism for each reaction below.
Short Answer
The product prediction and its mechanism for each of the reaction can be explained.
Learning Materials
Features
Discover
Chapter 20: Q24E (page 678)
Predict the product (s) and provide the mechanism for each reaction below.
The product prediction and its mechanism for each of the reaction can be explained.
All the tools & learning materials you need for study success - in one app.
Get started for free
The of p-cyclopropylbenzoic acid is 4.45. Is cyclopropylbenzene likely to be more reactive or less reactive than benzene toward electrophilic bromination? Explain.
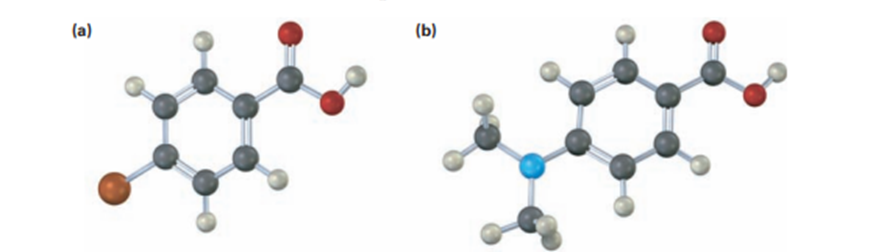
would you expect the following carboxylic acid to be more acidic or less acidic than benzoic acid? Explain (Reddish brown=Br)
Dicarboxylic acids have two dissociation constants, one for the initial dissociation into a monoanion and one for the second dissociation into a dianion. For oxalic acid, the first ionization constant is and the second ionization constant is . Why is the second carboxyl group far less acidic than the first?
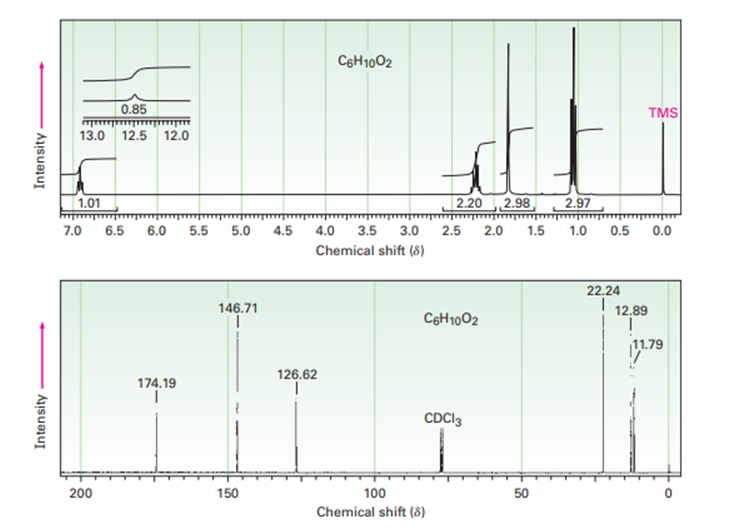
The 1H and 13C NMR spectra below belong to a compouns with formula C6H10O2. Propose a structure for this compound.
Assume you have a mixture of naphthalene and benzoic acid that you want to separate. How might you take advantage of the acidity of one component in the mixture to effect a separation?
What do you think about this solution?
We value your feedback to improve our textbook solutions.