Chapter 20: Q18E (page 653)
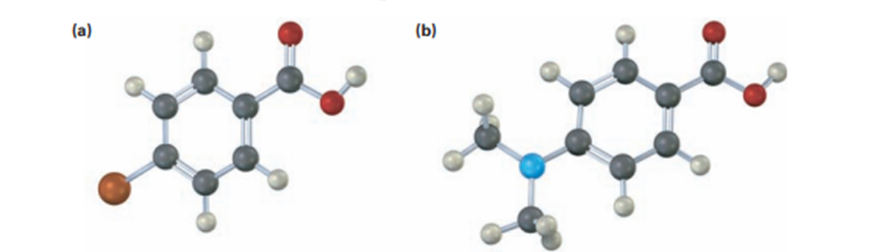
would you expect the following carboxylic acid to be more acidic or less acidic than benzoic acid? Explain (Reddish brown=Br)
Short Answer
To check whether the carboxylic acid is more or less acidic than benzoic acid.
Learning Materials
Features
Discover
Chapter 20: Q18E (page 653)
would you expect the following carboxylic acid to be more acidic or less acidic than benzoic acid? Explain (Reddish brown=Br)

To check whether the carboxylic acid is more or less acidic than benzoic acid.
All the tools & learning materials you need for study success - in one app.
Get started for free
Arrange the compounds in each of the following sets in order of increasing basicity:
(a) Magnesium acetate, magnesium hydroxide, methyl magnesium bromide
(b) Sodium benzoate, sodium p-nitrobenzoate, sodium acetylide
(c) Lithium hydroxide, lithium ethoxide, lithium formate
How would you prepare the following compounds from benzene? More than one step is required in each case.
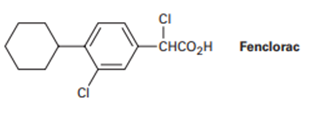
Propose a synthesis of the anti-inflammatory drug Fenclorac from phenyl cyclohexane.
How could you convert butanenitrile into the following compounds?
Write each step showing the reagents needed.
(a) 1-Butanol
(b) Butylamine
(c) 2-Methyl-3-hexanone
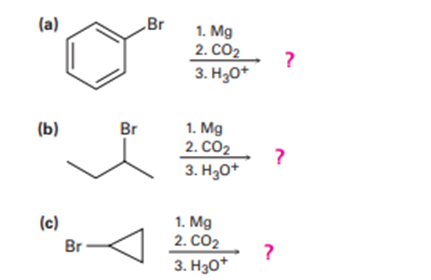
Predict the product and provide the mechanism for each reaction below.
What do you think about this solution?
We value your feedback to improve our textbook solutions.