Chapter 20: Q17E (page 653)
Give IUPAC names for the following carboxylic acids (reddish brown = Br);
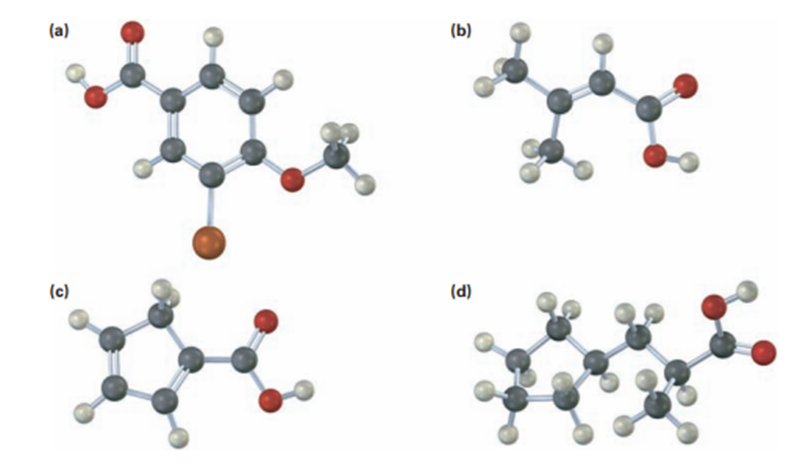
Short Answer
The IUPAC names of the given compounds can be found.
Learning Materials
Features
Discover
Chapter 20: Q17E (page 653)
Give IUPAC names for the following carboxylic acids (reddish brown = Br);

The IUPAC names of the given compounds can be found.
All the tools & learning materials you need for study success - in one app.
Get started for free
Acid-catalyzed hydrolysis of a nitrile to give a carboxylic acid occurs by initial protonation of the nitrogen atom, followed by nucleophilic addition of water. Review the mechanism of base-catalyzed nitrile hydrolysis in Section 20-7 and then predict the products for each reaction below and write all of the steps involved in the acid-catalyzed reaction, using curved arrows to represent electron flow in each step.
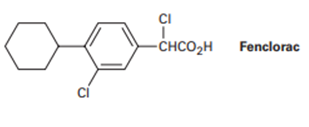
Propose a synthesis of the anti-inflammatory drug Fenclorac from phenyl cyclohexane.
Draw structures corresponding to the following IUPAC names:
Hepatanedioic acid
2-Hexen-4-ynoic acid
4-Ethyl-2-propyloctanoic acid
3-chlorophthalic acid
Triphenylacetic acid
2-Cyclobutenecarbonitrile
m-Benzoyl benzonitrile
In humans, the final product of purine degradation from DNA is uric acid, pKa = 5.61, which is excreted in the urine. What is the percent dissociation of uric acid in urine at a typical pH = 6.0? Why do you think uric acid is acidic even though it does not have a CO₂H group?
Predict the product and provide the mechanism for each reaction below.
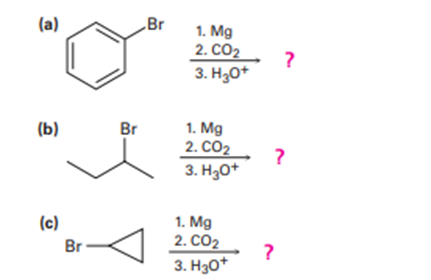
What do you think about this solution?
We value your feedback to improve our textbook solutions.