Chapter 20: 63E (page 678)
How would you carry out the following transformations? More than one step is needed in each case.
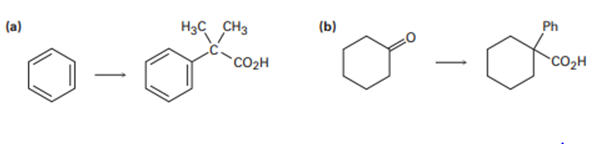
Short Answer
The transformation for the below structures has been shown.
Learning Materials
Features
Discover
Chapter 20: 63E (page 678)
How would you carry out the following transformations? More than one step is needed in each case.

The transformation for the below structures has been shown.
All the tools & learning materials you need for study success - in one app.
Get started for free
Naturally occurring compounds called terpenoids, which we’ll discuss in section27-5, are biosynthesized by a pathway that involves loss of carbon dioxide from 3-phosphomevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this reaction.
p-Aminobenzoic acid (PABA) is widely used as a sunscreen agent. Propose a synthesis of PABA starting from toluene.
How could you convert butanoic acid into the following compounds?
Write each step showing the reagents needed.
(a) 1-Butanol (b) 1-Bromobutane (c) Pentanoic acid
(d) 1-Butene (e) Octane
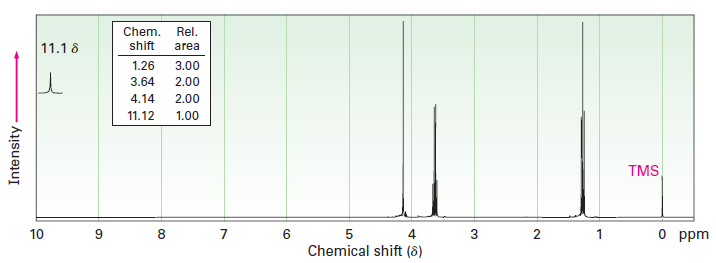
Compound A, C4H8O3, has infra-red absorptions at 1710 and 2500 to 3100 cm-1 and has the 1H NMR spectrum shown. Propose a structure for A.
The of p-cyclopropylbenzoic acid is 4.45. Is cyclopropylbenzene likely to be more reactive or less reactive than benzene toward electrophilic bromination? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.