Chapter 20: Q14E (page 653)
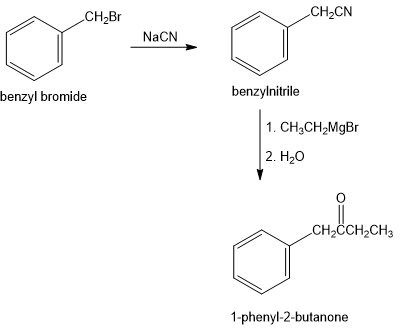
How would you prepare 1-phenyl-2-butanone, , from benzyl bromide, ? More than one step is required.
Short Answer
The preparation of 1-phenyl-2-butanone from benzyl bromide is shown in two step as follows:
Learning Materials
Features
Discover
Chapter 20: Q14E (page 653)
How would you prepare 1-phenyl-2-butanone, , from benzyl bromide, ? More than one step is required.
The preparation of 1-phenyl-2-butanone from benzyl bromide is shown in two step as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following carboxylic acids?
(a)
(b)
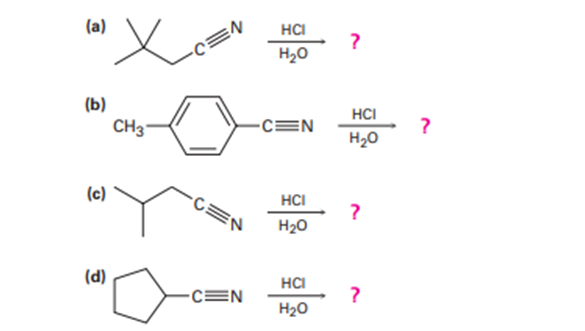
Acid-catalyzed hydrolysis of a nitrile to give a carboxylic acid occursby initial protonation of the nitrogen atom, followed by nucleophilicaddition of water. Review the mechanism of base-catalyzed nitrilehydrolysis in Section 20-7 and then predict the products for each reaction below and write all of the steps involved in the acid-catalyzedreaction, using curved arrows to represent electron flow in each step.
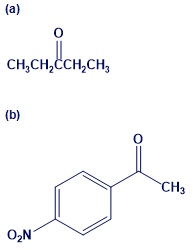
How would you prepare the following carbonyl compounds from a nitrile?
How could you convert butanenitrile into the following compounds?
Write each step showing the reagents needed.
(a) 1-Butanol
(b) Butylamine
(c) 2-Methyl-3-hexanone
How might you carry out the following transformation? More than one step is needed.
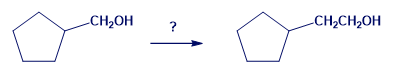
What do you think about this solution?
We value your feedback to improve our textbook solutions.