Chapter 21: Q-21-21-13P (page 701)
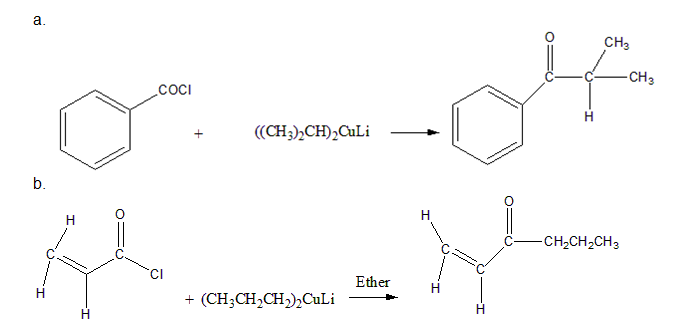
How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper reagent?
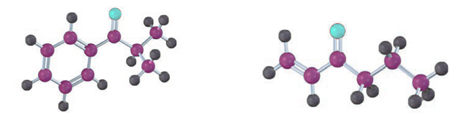
Short Answer
Learning Materials
Features
Discover
Chapter 21: Q-21-21-13P (page 701)
How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper reagent?


All the tools & learning materials you need for study success - in one app.
Get started for free
21-51 What product would you expect to obtain from Grignard reaction ofan excess of phenylmagnesium bromide with dimethyl carbonate, ?
The following structure represents a tetrahedral alkoxide ion intermediate formed by the addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the starting acid derivative, and the ultimate product.
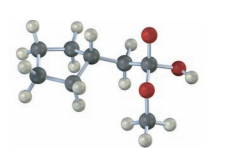
How would you convert N-ethylbenzamide to each of the following products?
(a) Benzoic acid (b) Benzyl alcohol (c)
Draw structures corresponding to the following names:
(a) Phenyl benzoate
(b) N-Ethyl-N-methylbutanamide
(c) 2,4-Dimethylpentanoyl chloride
(d) Methyl 1-methylcyclohexanecarboxylate
(e) Ethyl 3-oxopentanoate
(f) Methyl p-bromobenzenethioate
(g) Formic propanoic anhydride
(h) cis-2-Methylcyclopentanecarbonyl bromide
21-48 Predict the product, if any, of the reaction between propanoyl chloride andthe following reagents:
What do you think about this solution?
We value your feedback to improve our textbook solutions.