Chapter 21: Q-21-21-5P (page 688)
Predict the products of the following nucleophilic acyl substitution reactions:
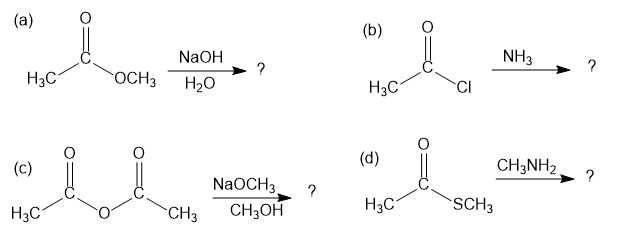
Short Answer
The products of each reaction are as shown below:
Learning Materials
Features
Discover
Chapter 21: Q-21-21-5P (page 688)
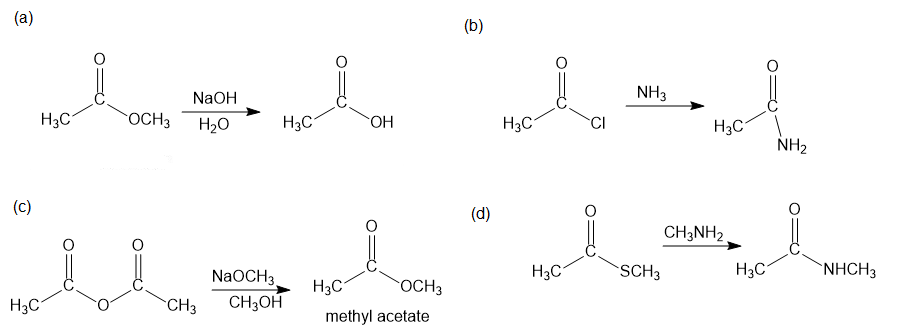
Predict the products of the following nucleophilic acyl substitution reactions:

The products of each reaction are as shown below:

All the tools & learning materials you need for study success - in one app.
Get started for free
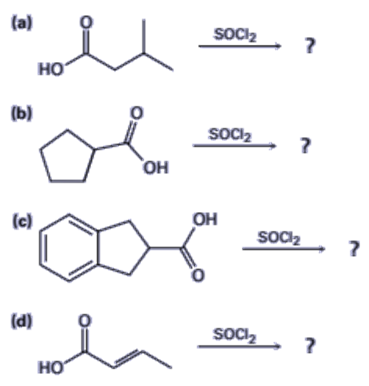
21-32 Predict the product(s) and provide the mechanism for each reactionbelow.
Write the mechanism of the reaction shown in Figure 21-9 between coenzyme A and acetyl adenylate to give acetyl CoA.
How might you prepare the following esters using a nucleophilic acyl substitution reaction of an acid chloride?
(a)
(b)
(c) Ethyl benzoate.
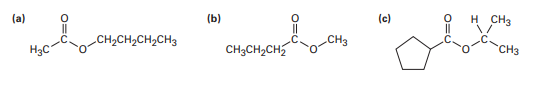
How might you prepare the following esters from the corresponding acids?
21-70 Treatment of a carboxylic acid with trifluoroacetic anhydride leads to anunsymmetrical anhydride that rapidly reacts with alcohol to give an ester.
(a) Propose a mechanism for the formation of the unsymmetrical anhydride.
(b) Why is the unsymmetrical anhydride unusually reactive?
(c) Why does the unsymmetrical anhydride react as indicated ratherthan giving a trifluoroacetate ester plus carboxylic acid?
What do you think about this solution?
We value your feedback to improve our textbook solutions.