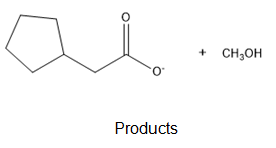
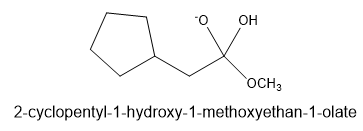
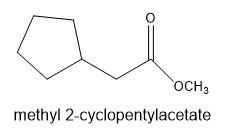
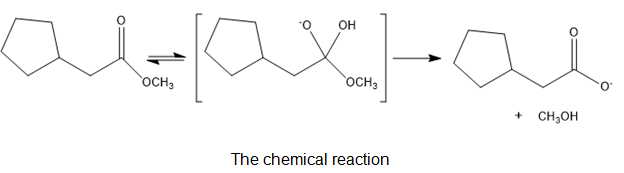
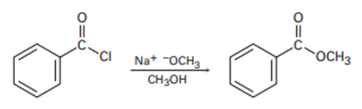
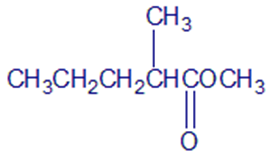
Chapter 21: Q-21-21-6P (page 688)
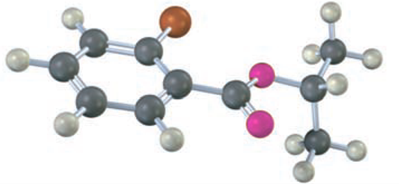
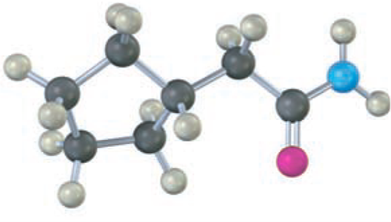
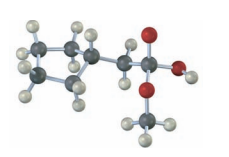
The following structure represents a tetrahedral alkoxide ion intermediate formed by the addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the starting acid derivative, and the ultimate product.
Short Answer
The nucleophile is .
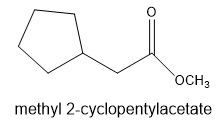
The leaving group in the molecule is . The derivative of starting acid is methyl-2-cyclopentylacetate.
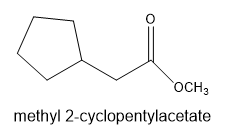
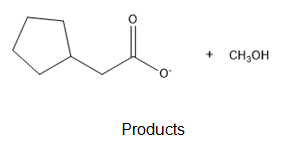
The ultimate products are methanol and cyclopentylacetate anion.