Chapter 21: Q-21-21-12P (page 699)
Question: How could you prepare the following amides using an acid chloride and an amine or ammonia?
- N,N-Dimethylbenzamide
- Propanamide
Short Answer
(a)
(b)
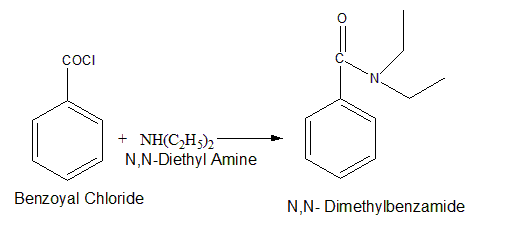
(c)
Learning Materials
Features
Discover
Chapter 21: Q-21-21-12P (page 699)
Question: How could you prepare the following amides using an acid chloride and an amine or ammonia?
(a)
(b)

(c)
All the tools & learning materials you need for study success - in one app.
Get started for free
21-72 Phenyl 4-aminosalicylate is a drug used in the treatment of tuberculosis. Propose a synthesis of this compound starting from 4-nitrosalicylic acid.
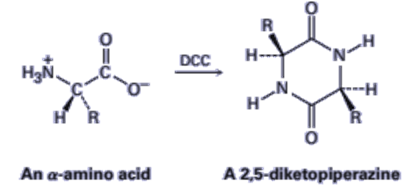
21-38 Treatment of an a-amino acid with DCC yields a 2,5-diketopiperazine.Propose a mechanism.
21-36 When 4-dimethylaminopyridine (DMAP) is added in catalytic amountsto acetic anhydride and an alcohol, it significantly increases the rate of ester formation. The process begins with a reaction between acetic anhydride and DMAP to form a highly reactive acetylpyridinium intermediate that is more reactive than acetic anhydride itself. Propose a mechanism for this process that includes the formation and reaction of the acetylpyridinium intermediate.
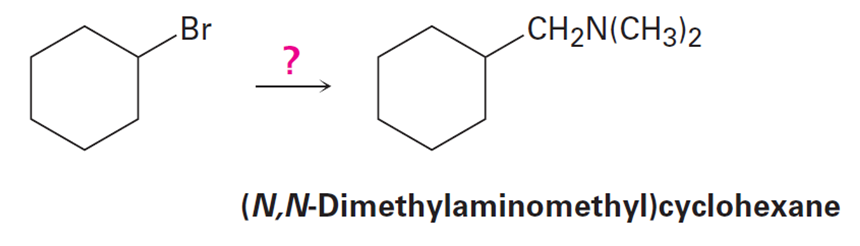
How would you use the reaction of an amide with LiAlH4 as the key step in going from bromocyclohexane to (N,N-dimethylaminomethyl)cyclohexane? Write all the steps in the reaction sequence.
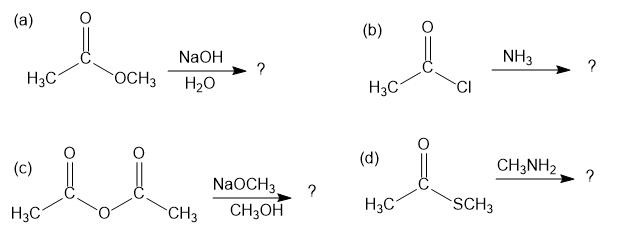
Predict the products of the following nucleophilic acyl substitution reactions:
What do you think about this solution?
We value your feedback to improve our textbook solutions.