Chapter 22: 9P (page 728)
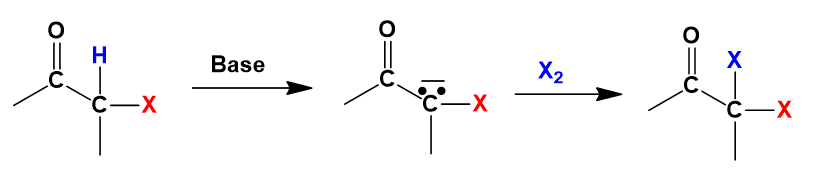
Why do you suppose ketone halogenations in acidic media are referred to as being acid-catalyzed,whereas halogenations in basic media are base-promoted? In other words, why is a full equivalent of base required for halogenation?
Short Answer
You need to explain why a full equivalent of base required for halogenation of ketone.