Chapter 6: An Overview of Organic Reactions
Q20E
Draw an energy diagram for a one-step reaction with . Label the parts of the diagram corresponding to reactants, products, transition state, , and . Is positive or negative?
Q21E
Draw an energy diagram for a two-step reaction with > 1. Label the overall , transition states, and intermediate. Is positive or negative?
Q22E
Draw an energy diagram for a two-step exergonic reaction whose second step is faster than its first step.
Q23E
Draw an energy diagram for a reaction with . What is the value of in this reaction?
Q24E
The addition of water to ethylene to yield ethanol has the following thermodynamic parameters:
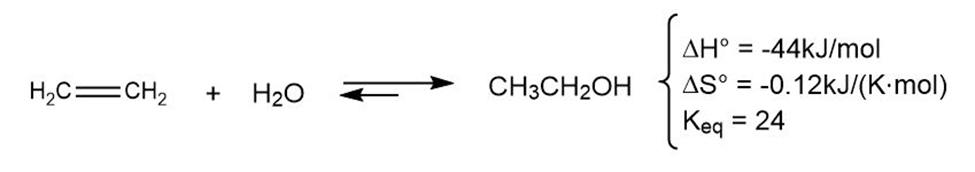
(a) Is the reaction exothermic or endothermic?
(b) Is the reaction favorable (spontaneous) or unfavorable (nonspontaneous) at room temperature (298 K)?
Q25E
When isopropylidenecyclohexane is treated with strong acid at room
temperature, isomerization occurs by the mechanism shown below to yield 1-isopropylcyclohexene:
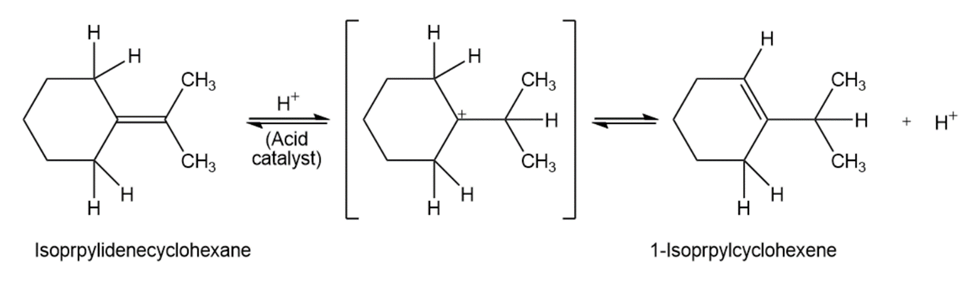
At equilibrium, the product mixture contains about 30% isopropylidene- cyclohexane and about 70% 1-isopropylcyclohexene.
(a)What is an approximate value of Keq for the reaction?
(b)Since the reaction occurs slowly at room temperature, what is its approximateG‡?
(c)Draw an energy diagram for the reaction.
Q27E
Draw the electron-pushing mechanism for each radical reaction below. Identify each step as initiation, propagation, or termination.
(a)
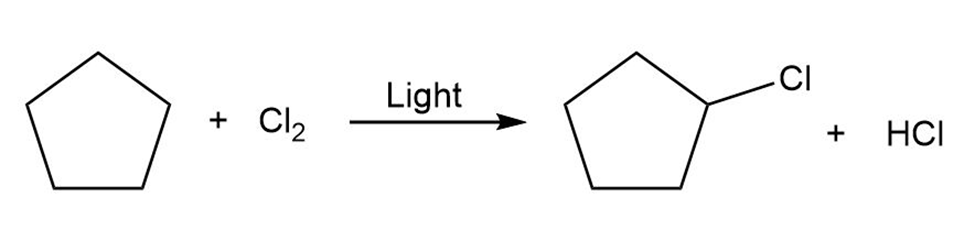
b)
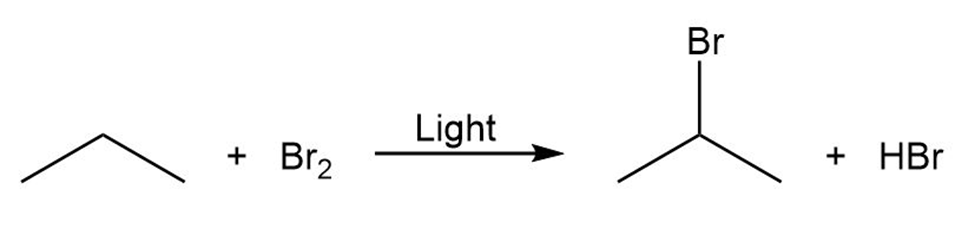
c)
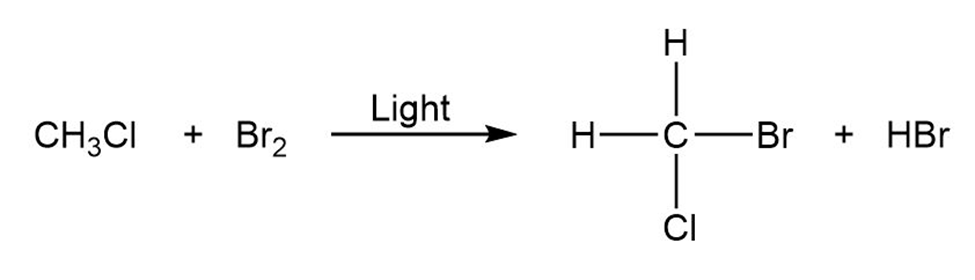
Q28E
Draw the complete mechanism for each polar reaction below.
(a)
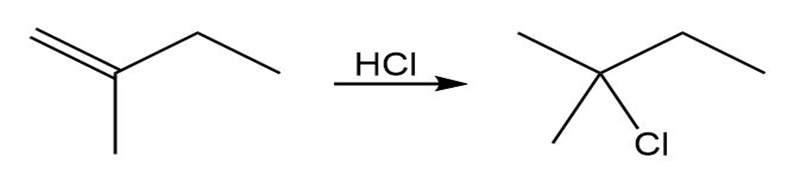
(b)
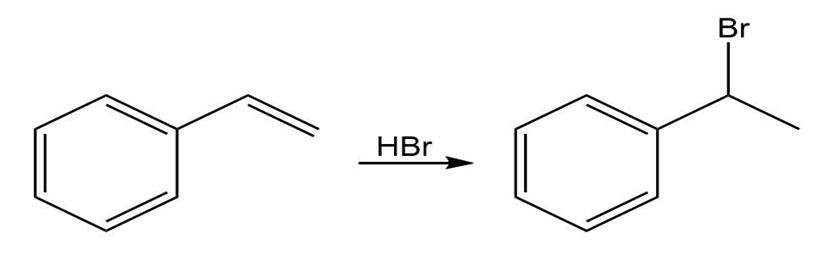
(c)
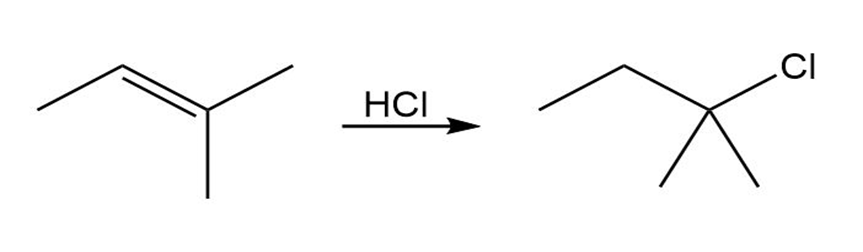
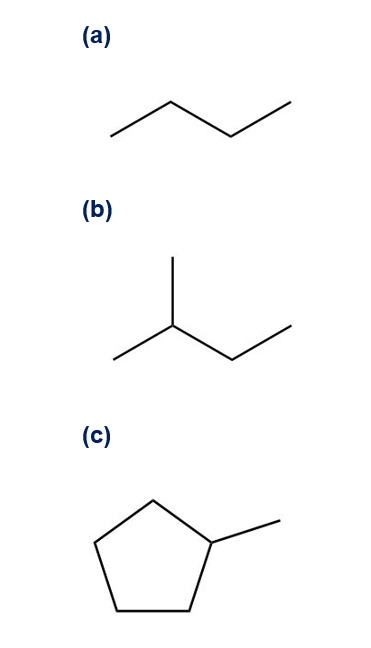
Q38E
Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated compounds. For which of the following compounds does radical chlorination give a single monochloro product?
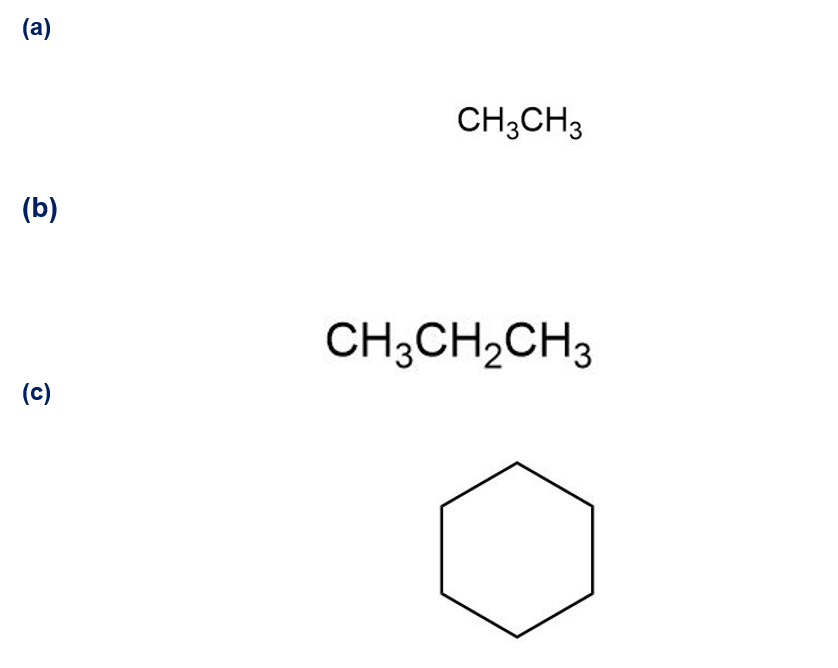
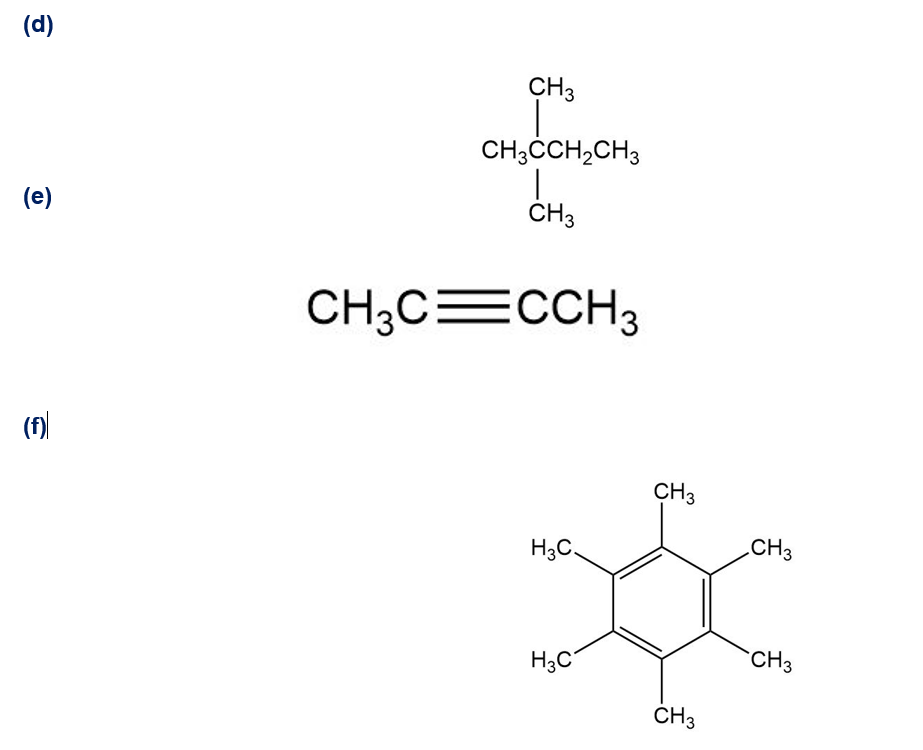
Q39E
Draw all of the different monochlorinated products one would obtain by the radical chlorination of these compounds. (Do not consider the stereochemistry of the products in your answer.)