Chapter 6: Q28E (page 181)
Draw the complete mechanism for each polar reaction below.
(a)
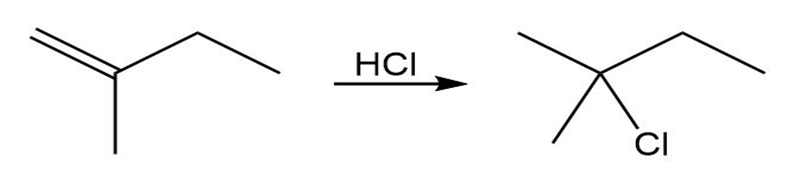
(b)
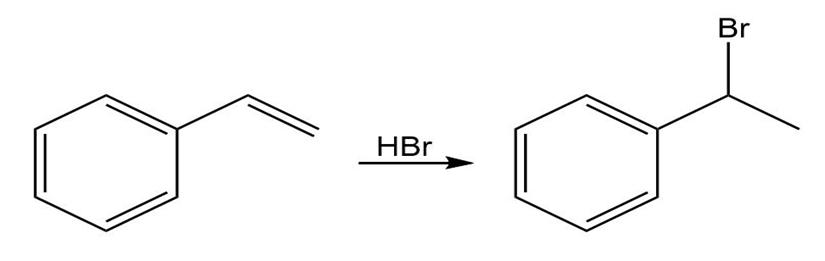
(c)
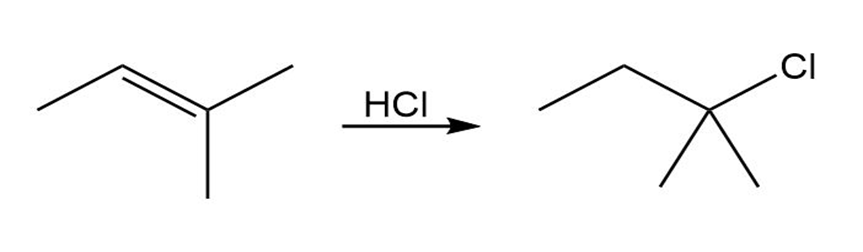
Short Answer
(a)
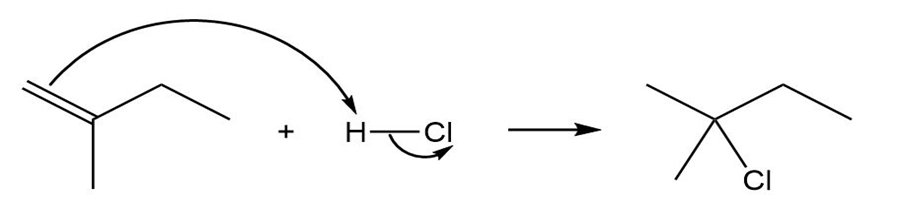
(b)
(c)
Learning Materials
Features
Discover
Chapter 6: Q28E (page 181)
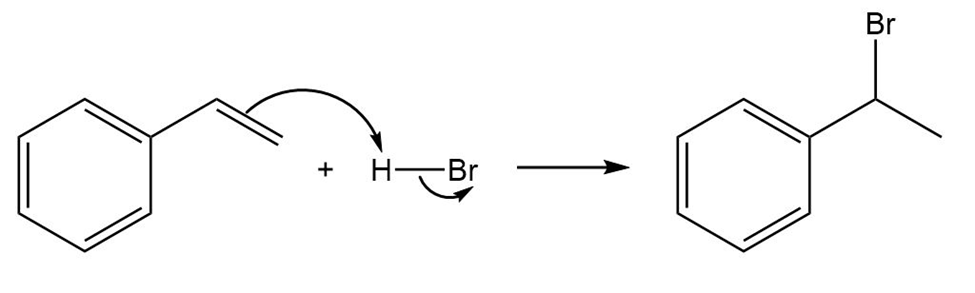
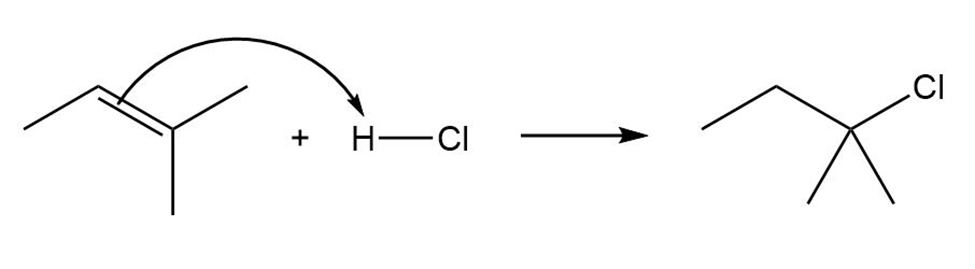
Draw the complete mechanism for each polar reaction below.
(a)

(b)

(c)

(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
When isopropylidenecyclohexane is treated with strong acid at room
temperature, isomerization occurs by the mechanism shown below to yield 1-isopropylcyclohexene:
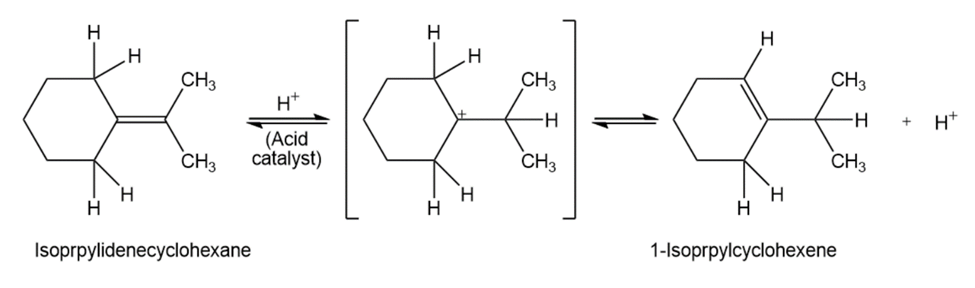
At equilibrium, the product mixture contains about 30% isopropylidene- cyclohexane and about 70% 1-isopropylcyclohexene.
(a)What is an approximate value of Keq for the reaction?
(b)Since the reaction occurs slowly at room temperature, what is its approximateG‡?
(c)Draw an energy diagram for the reaction.
Draw an energy diagram for a two-step exergonic reaction whose second step is faster than its first step.
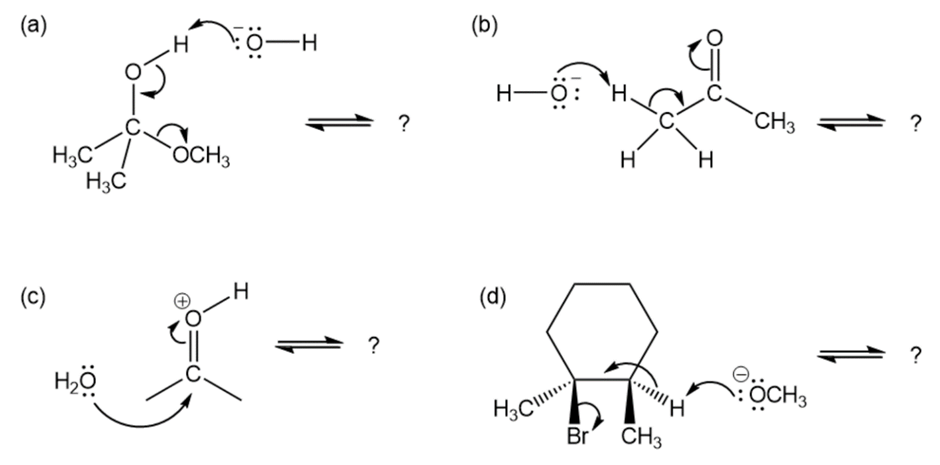
Follow the flow of electrons indicated by the curved arrows in each of the following polar reactions, and predict the products that result:
Draw an energy diagram for a two-step reaction with > 1. Label the overall , transition states, and intermediate. Is positive or negative?
Draw an energy diagram for a reaction with . What is the value of in this reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.