Chapter 7: Q7-56E (page 219)
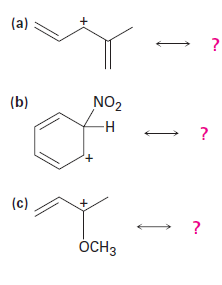
Carbocations can be stabilized by resonance. Draw all the resonance forms that would stabilize each carbocation:
Short Answer
.
Learning Materials
Features
Discover
Chapter 7: Q7-56E (page 219)
Carbocations can be stabilized by resonance. Draw all the resonance forms that would stabilize each carbocation:

.
All the tools & learning materials you need for study success - in one app.
Get started for free
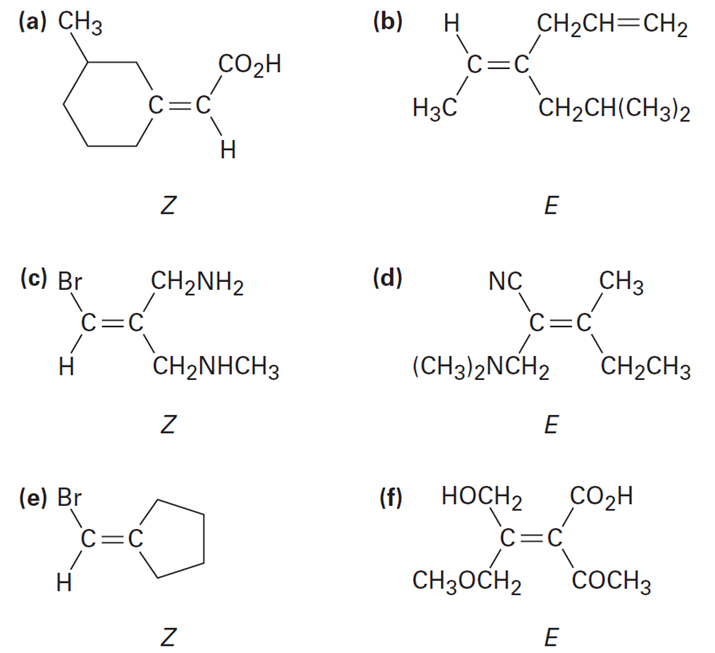
Question : Which of the following E, Z designations are correct, and which are incorrect ?
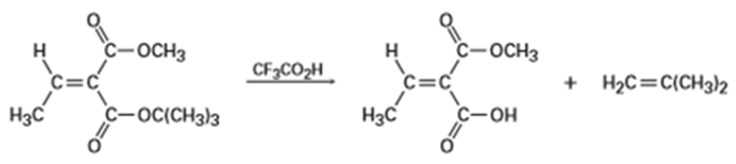
tert-Butyl [RCO2C(CH3)3] esters are converted into carboxylic acids [RC02H] by reaction with trifluoroacetic acid, a reaction useful in protein synthesis (Section 26-7). Assign E, Z designation to the double bonds of both reactant and product in the following scheme, and explain why there is an apparent change in double-bond stereochemistry:
Which member in each of the following sets ranks higher?
Trans-Cyclooctene is less stable than cis-cyclooctene by 38.5 kJ/mol, but trans-cyclononene is less stable than cis-cyclononene by only 12.2 kJ/mol. Explain.
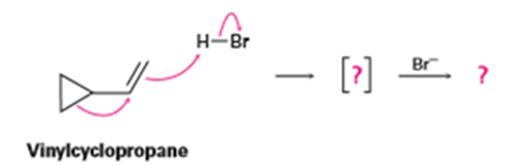
Vinylcyclopropane reacts with HBr to yield a rearranged alkyl bromide. Follow the flow of electrons as represented by the curved arrows, show the structure of the carbocation intermediate in brackets, and show the structure of the final product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.