Chapter 7: Q7-55E (page 219)
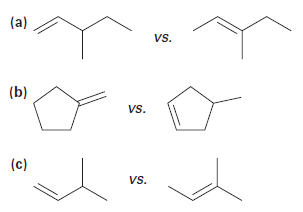
Use Hammond’s Postulate to determine which alkene in each pair would be expected to form a carbocation faster in an electrophilic addition reaction:
Short Answer
.
Learning Materials
Features
Discover
Chapter 7: Q7-55E (page 219)
Use Hammond’s Postulate to determine which alkene in each pair would be expected to form a carbocation faster in an electrophilic addition reaction:

.
All the tools & learning materials you need for study success - in one app.
Get started for free
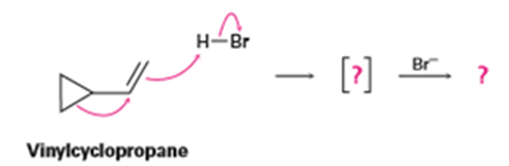
Vinylcyclopropane reacts with HBr to yield a rearranged alkyl bromide. Follow the flow of electrons as represented by the curved arrows, show the structure of the carbocation intermediate in brackets, and show the structure of the final product.
Draw structures corresponding to the following IUPAC names:
Q 44Draw and name the 17 alkene isomers,C6H12 , including E, Z isomers.
Question: Which member in each of the following sets ranks higher?
Rank the substituents in each of the following sets according to the sequence rules:
What do you think about this solution?
We value your feedback to improve our textbook solutions.