Chapter 7: Q12P-c (page 197)
Rank the substituents in each of the following sets according to the sequence rules:
Short Answer
Higher to lower-ranking:
Learning Materials
Features
Discover
Chapter 7: Q12P-c (page 197)
Rank the substituents in each of the following sets according to the sequence rules:
Higher to lower-ranking:
All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Draw and name the six alkene isomers, C5H10 , including E, Zisomers:
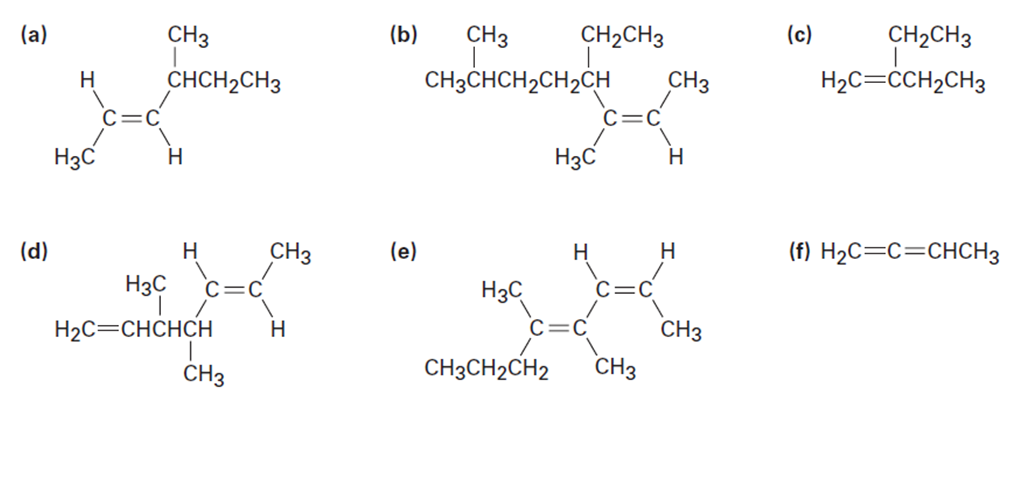
Name the following alkenes:
Draw an energy diagram for the addition of HBr to 1-pentene. Let one curve on your diagram show the formation of 1-bromopentane product and another curve on the same diagram show the formation of 2-bromopentane product. Label the positions for all reactants, intermediates, and products. Which curve has the higher-energy carbocation intermediate? Which curve has the higher-energy first transition state?
Rank the substituents in each of the following sets according to the sequence rules:
What about the second step in the electrophilic addition of HCl to an alkene—the reaction of chloride ion with the carbocation intermediate? Is this step exergonic or endergonic? Does the transition state for this second step resemble the reactant (carbocation) or product (alkyl chloride)? Make a rough drawing of what the transition-state structure might look like.
What do you think about this solution?
We value your feedback to improve our textbook solutions.