Chapter 7: Q12P-a (page 197)
Rank the substituents in each of the following sets according to the sequence rules:
Short Answer
Higher to lower-ranking:
Learning Materials
Features
Discover
Chapter 7: Q12P-a (page 197)
Rank the substituents in each of the following sets according to the sequence rules:
Higher to lower-ranking:
All the tools & learning materials you need for study success - in one app.
Get started for free
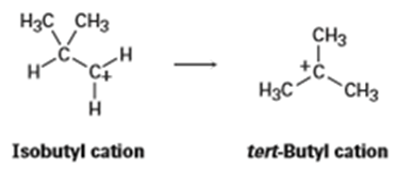
The isobutyl cation spontaneously rearranges to the tert-butyl cation by a hydride shift. Is the rearrangement exergonic or endergonic? Draw what you think the transition state for the hydride shift might look like according to the Hammond postulate.
Sketch the transition-state structures involved in the reaction of HBr with 1-pentene (Problem 7-69). Tell whether each structure resembles reactant or product.
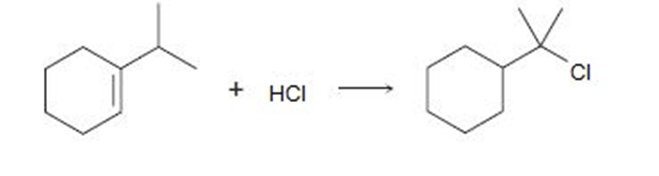
Question: Addition of HCl to 1-isopropylcyclohexene yields a rearranged product.
Propose a mechanism, showing the structures of the intermediates and using curved arrows to indicate electron flow in each step
Which member in each of the following sets ranks higher?
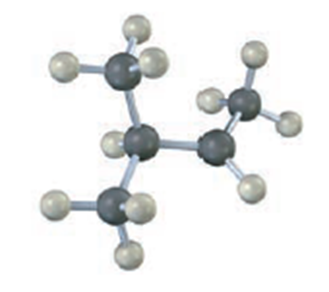
Draw a skeletal structure of the following carbocation. Identify it as primary, secondary, or tertiary, and identify the hydrogen atoms that have the proper orientation for hyperconjugation in the conformation shown.
What do you think about this solution?
We value your feedback to improve our textbook solutions.