Chapter 7: Q7-52E (page 219)
Trans-Cyclooctene is less stable than cis-cyclooctene by 38.5 kJ/mol, but trans-cyclononene is less stable than cis-cyclononene by only 12.2 kJ/mol. Explain.
Short Answer
a
Learning Materials
Features
Discover
Chapter 7: Q7-52E (page 219)
Trans-Cyclooctene is less stable than cis-cyclooctene by 38.5 kJ/mol, but trans-cyclononene is less stable than cis-cyclononene by only 12.2 kJ/mol. Explain.
a
All the tools & learning materials you need for study success - in one app.
Get started for free
What alkenes would you start with to prepare the following products?
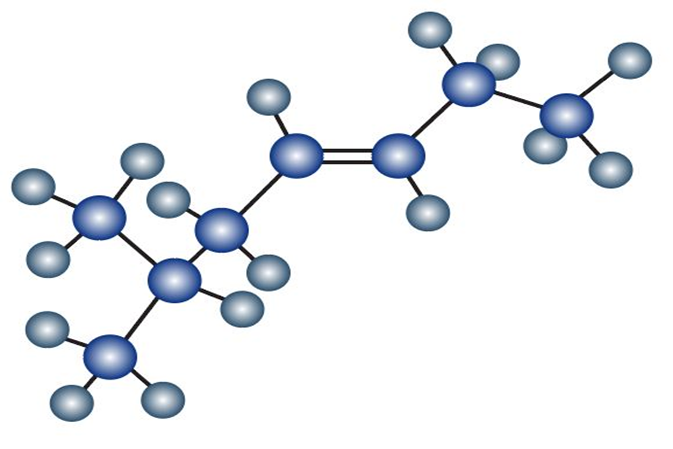
Allene (1,2-propadiene), H2C = C = CH2, has two adjacent double bonds. What kind of hybridization must the central carbon have? Sketch the bonding orbitals in allene. What shape do you predict for allene?
Which member in each of the following sets ranks higher?
Draw structures corresponding to the following systematic names:
(a)(4E)-2, 4-Dimethyl-1, 4-hexadiene
(b)cis-3, 3-Dimethyl-4-propyl-1, 5-octadiene
(c)4-Methyl-1, 2-pentadiene
(d)(3E,5Z)-2, 6-Dimethyl-1, 3, 5,7-octatetraene
(e)3-Butyl-2-heptene
(f)trans-2, 2, 5, 5-Tetramethyl-3-hexene
Question: Name the following alkenes, including a cis or trans designation:
What do you think about this solution?
We value your feedback to improve our textbook solutions.