Chapter 7: Q7-26E (page 219)
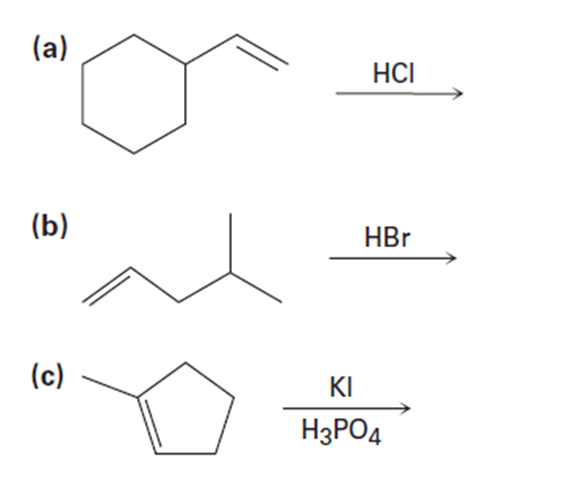
Predict the major product and show the complete mechanism for eachelectrophilic reaction below.
Short Answer
a)
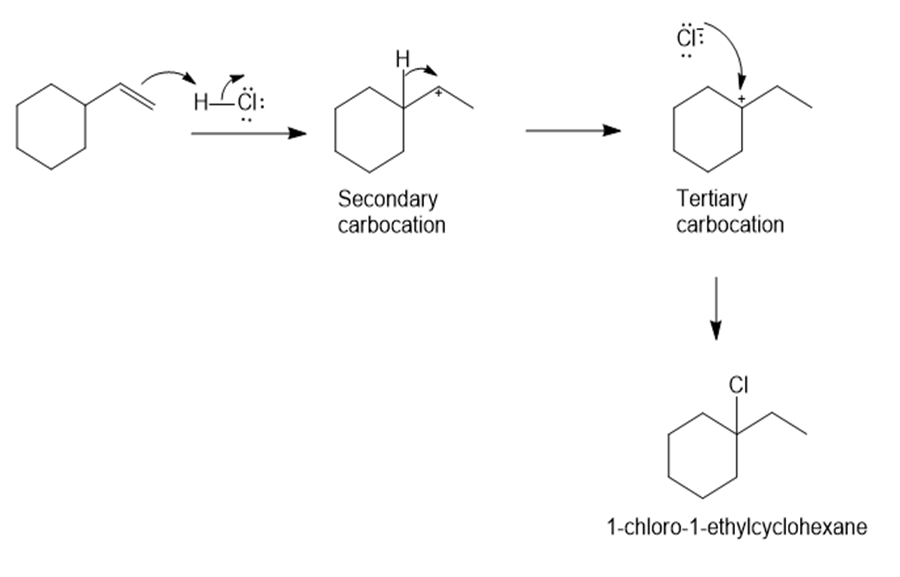
b)
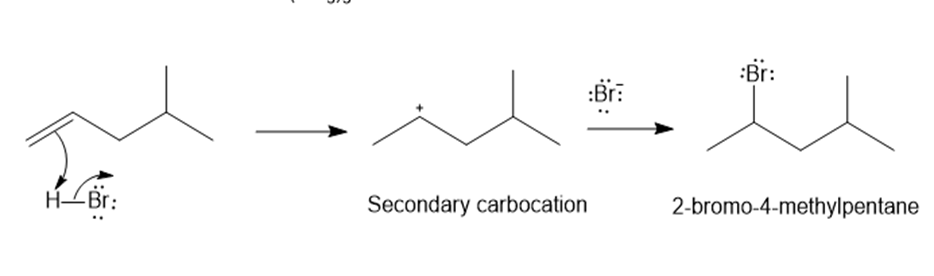
c)
Learning Materials
Features
Discover
Chapter 7: Q7-26E (page 219)
Predict the major product and show the complete mechanism for eachelectrophilic reaction below.

a)

b)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
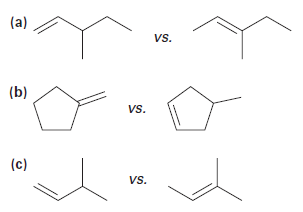
Use Hammond’s Postulate to determine which alkene in each pair would be expected to form a carbocation faster in an electrophilic addition reaction:
How many hydrogens does each of the following compounds have?
(a) , has two rings and one double bond
(b) , has two double bonds
(c) , has one ring and three double bonds
Sketch the transition-state structures involved in the reaction of HBr with 1-pentene (Problem 7-69). Tell whether each structure resembles reactant or product.
Which member in each of the following sets ranks higher?
Which of the following compounds can exist as pairs of cis-trans isomers? Draw each cis-trans pair, and indicate the geometry of each isomer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.