Chapter 7: Q7-25E (page 219)
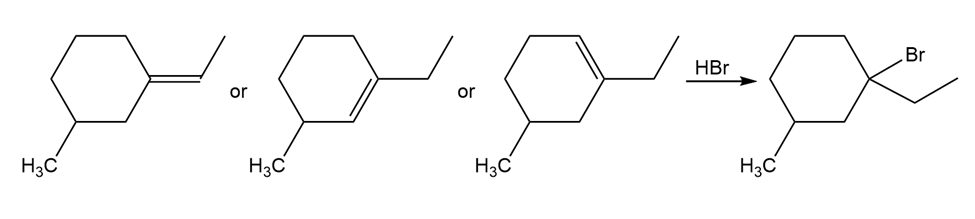
The following alkyl bromide can be made by HBr addition to three different alkenes. Show their structures.
Short Answer
Learning Materials
Features
Discover
Chapter 7: Q7-25E (page 219)
The following alkyl bromide can be made by HBr addition to three different alkenes. Show their structures.


All the tools & learning materials you need for study success - in one app.
Get started for free
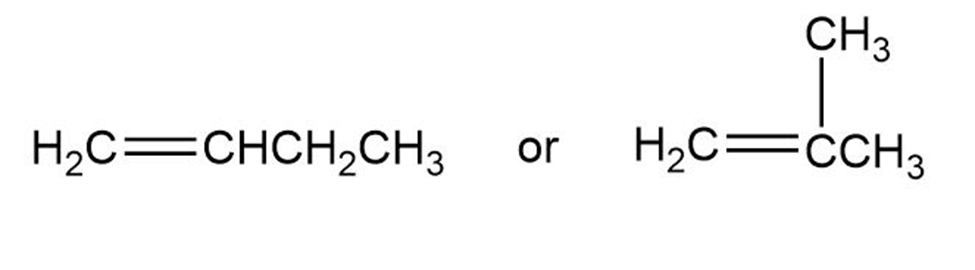
Name the following alkenes, and tell which compound in each pair is more stable:
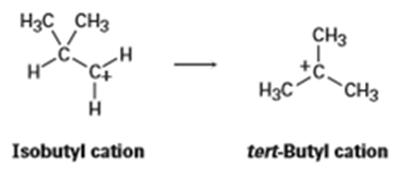
The isobutyl cation spontaneously rearranges to the tert-butyl cation by a hydride shift. Is the rearrangement exergonic or endergonic? Draw what you think the transition state for the hydride shift might look like according to the Hammond postulate.
Draw structures corresponding to the following systematic names:
(a)(4E)-2, 4-Dimethyl-1, 4-hexadiene
(b)cis-3, 3-Dimethyl-4-propyl-1, 5-octadiene
(c)4-Methyl-1, 2-pentadiene
(d)(3E,5Z)-2, 6-Dimethyl-1, 3, 5,7-octatetraene
(e)3-Butyl-2-heptene
(f)trans-2, 2, 5, 5-Tetramethyl-3-hexene
Question: Calculate the degree of unsaturation in the following formulas, and
draw five possible structures for each:
Rank the substituents in each of the following sets according to the sequence rules:
What do you think about this solution?
We value your feedback to improve our textbook solutions.