Chapter 19: Q15P (page 629)
Identify the carbonyl compound and the alcohol that were used to prepare the following acetal:
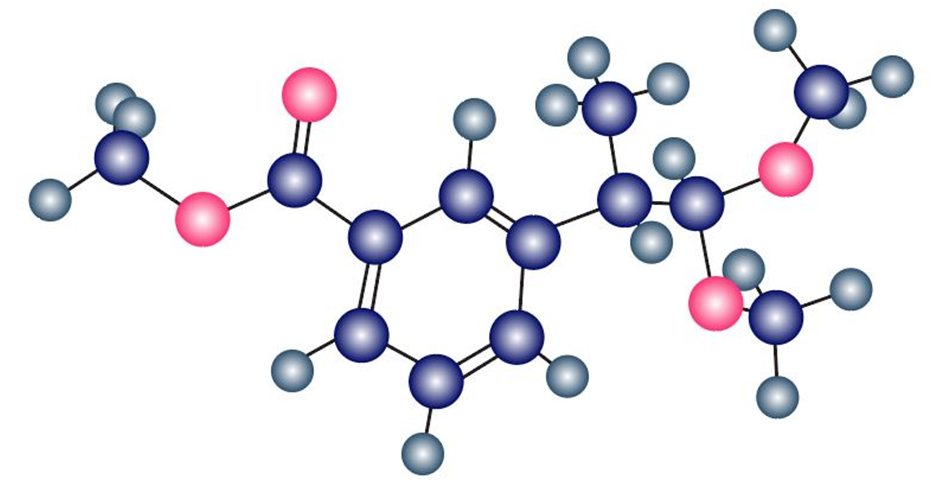
Short Answer
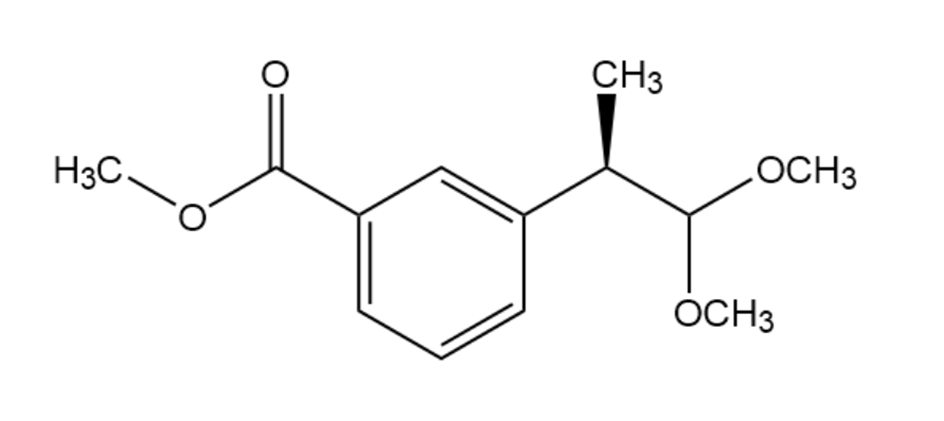
Skeletal structure
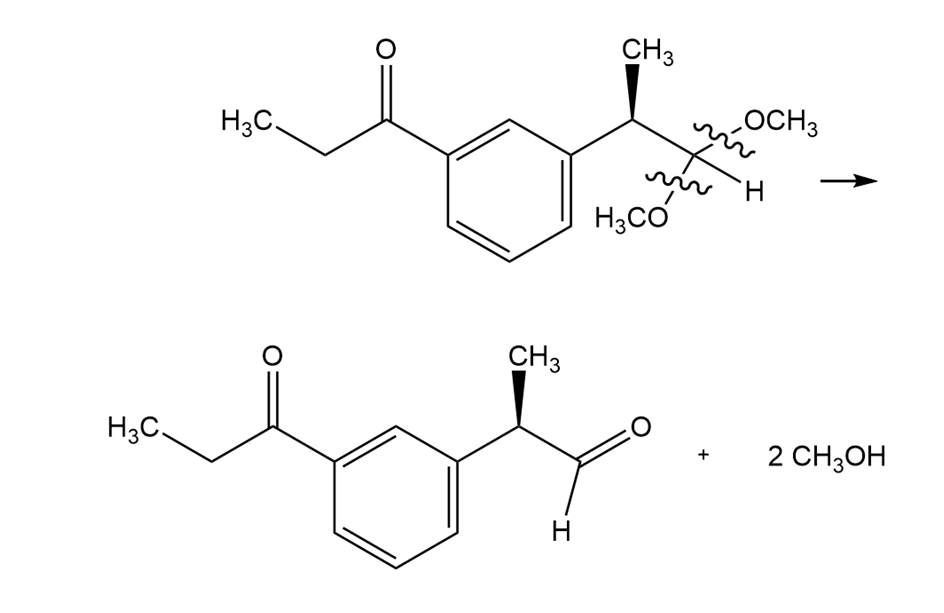
Formation of starting materials
Learning Materials
Features
Discover
Chapter 19: Q15P (page 629)
Identify the carbonyl compound and the alcohol that were used to prepare the following acetal:


Skeletal structure

Formation of starting materials
All the tools & learning materials you need for study success - in one app.
Get started for free
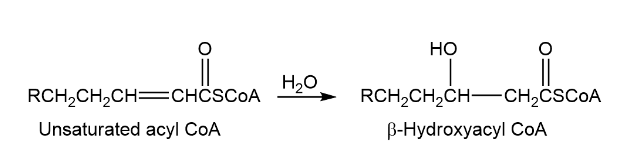
One of the steps in metabolism of fats is the reaction of an unsaturated acyl coA with water to give a - hydroxyacyl CoA. Propose a mechanism.
Treatment of an aldehyde or ketone with cyanide ion (), followed by protonation of the tetrahedral alkoxide ion intermediate, gives a cyanohydrin.
Show the structure of the cyanohydrin obtained from cyclohexanone.
When dissolved in water, trichloroacetaldehyde exists primarily as its hydrate, called chloral hydrate. Show the structure of chloral hydrate.
Reaction of 2-butanone with HCN yields a chiral product. What stereochemistry does the product have? Is it optically active?
Compound B is isomeric with A (Problem 19-79) and shows an IR peak at 1715 cm-1. The 1HNMR spectrum of B has peaks at 2.4 (1 H, septet, J = 7 Hz), 2.1 (3 H, singlet), and 1.2 (6 H, doublet, J =7 Hz). What is
the structure of B?
What do you think about this solution?
We value your feedback to improve our textbook solutions.