Chapter 6: Problem 6.28 (page 241)
Classify each transformation as substitution, elimination, or addition.
a.
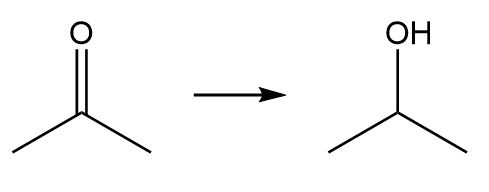
b.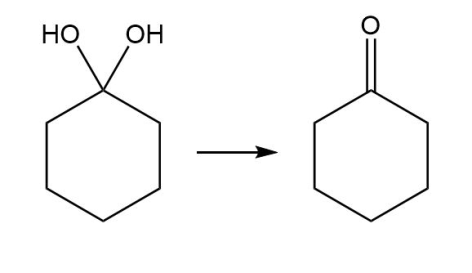
Short Answer
Answer
- Elimination reaction
- Addition reaction
Learning Materials
Features
Discover
Chapter 6: Problem 6.28 (page 241)
Classify each transformation as substitution, elimination, or addition.
a.

b.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Which value corresponds to a negative value of or ?
b. In a unimolecular reaction with five times as much starting material as product at equilibrium, what is the value of Keq? Is positive or negative?
c. Which value corresponds to a larger : or ?
Label each statement as true or false. Correct any false statement to make it true.
a. Increasing temperature increases reaction rate.
b. If a reaction is fast, it has a large rate constant.
c. A fast reaction has a large negative value.
d. When is large, the rate constant k is also large.
e. Fast reactions have equilibrium constants > 1.
f. Increasing the concentration of a reactant always increases the rate of a reaction.
For a reaction with and , decide which of the following statements is (are) true. Correct any false statement to make it true. Ignore entropy considerations. (a) The reaction is faster than a reaction with and . (b) The reaction is faster than a reaction with and . (c) for the reaction is a positive value. (d) The starting materials are lower in energy than the products of the reaction. (e) The reaction is exothermic.
As we learned in Chapter 4, propane has both and hydrogens.
Identify the catalyst in each equation.
a.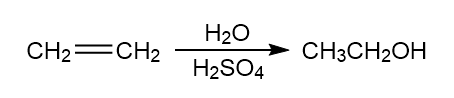
b.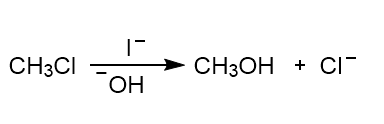
What do you think about this solution?
We value your feedback to improve our textbook solutions.