Chapter 6: Problem 6.29 (page 241)
Use full-headed or half-headed curved arrows to show the movement of electrons in each reaction.
a.
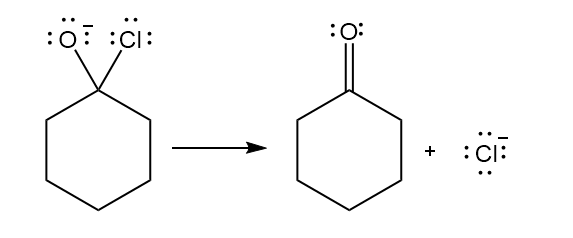
b.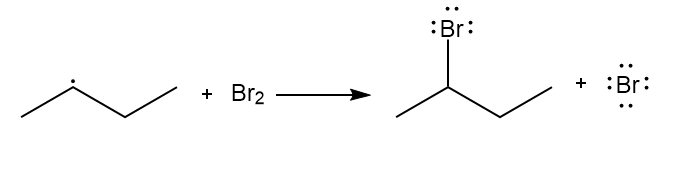
c.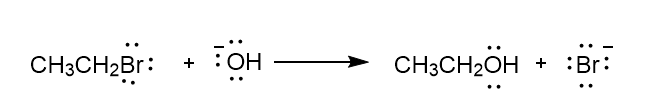
d.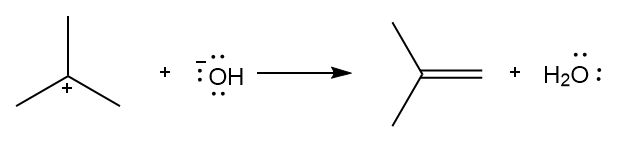
Short Answer
Answer
a.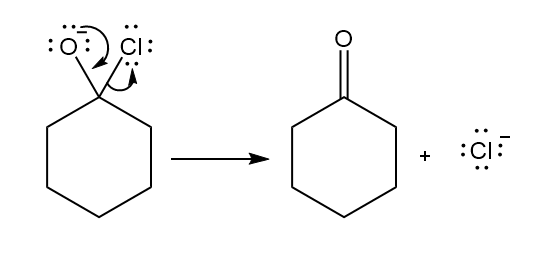
b.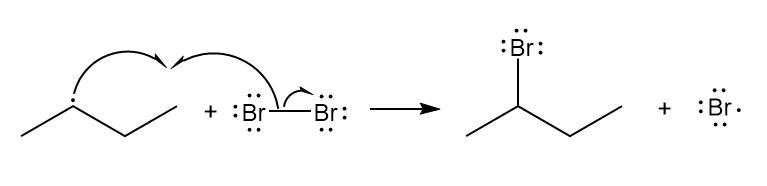
c.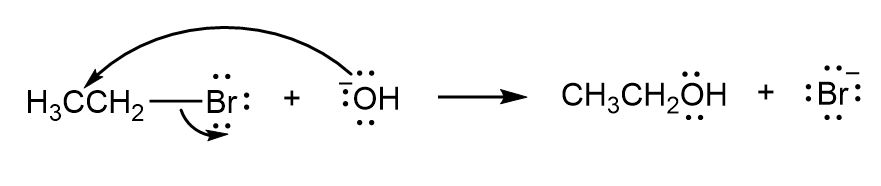
d.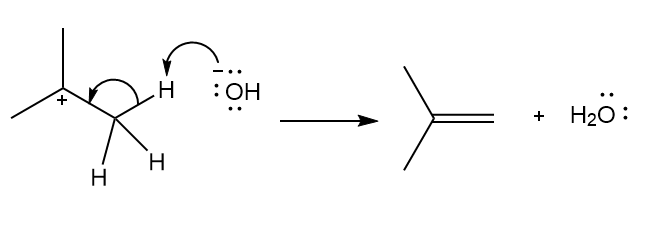
Learning Materials
Features
Discover
Chapter 6: Problem 6.29 (page 241)
Use full-headed or half-headed curved arrows to show the movement of electrons in each reaction.
a.

b.
c.
d.
Answer
a.
b.
c.
d.
All the tools & learning materials you need for study success - in one app.
Get started for free
For each rate equation, what effect does the indicated concentration change have on the overall rate of the reaction?
[1]
[2]role="math" localid="1648280223497"
Draw the products of each reaction by following the curved arrows.
a.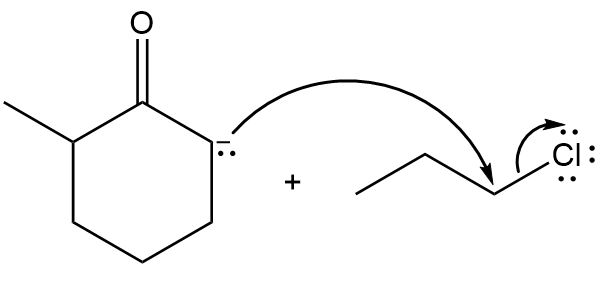
b.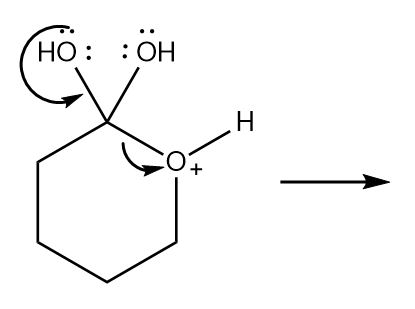
Given each value, determine whether the starting material or the product is favored at equilibrium.
a.
b.
c.
d.
e.
f.
g.
h.
Given each of the following values, is the starting material or product favored at equilibrium?
a.
b.
Indicate which factors affect the rate of a reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.