Chapter 6: Problem 6.48 (page 244)
Indicate which factors affect the rate of a reaction.
- Temperature
- concentration
- width="28">
K eq - k
- Catalysts
Short Answer
Answer
(c.), (d.), e(.), (g.), and (h.) affect the rate of the reaction.
Learning Materials
Features
Discover
Chapter 6: Problem 6.48 (page 244)
Indicate which factors affect the rate of a reaction.
Answer
(c.), (d.), e(.), (g.), and (h.) affect the rate of the reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
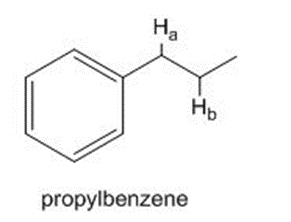
What carbon radical is formed by homolysis of the bond in propylbenzene? Draw all reasonable resonance structures for this radical.
What carbon radical is formed by homolysis of therole="math" localid="1648540916945" bond in propylbenzene? Draw all reasonable resonance structures for this radical.
The bond dissociation energy of one of the C-H bonds is considerably less than the bond dissociation energy of the other. Which C-H bond is weaker? Offer an explanation.
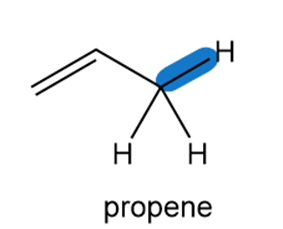
Homolysis of the indicated C-H bond in propene forms a resonance-stabilized radical.
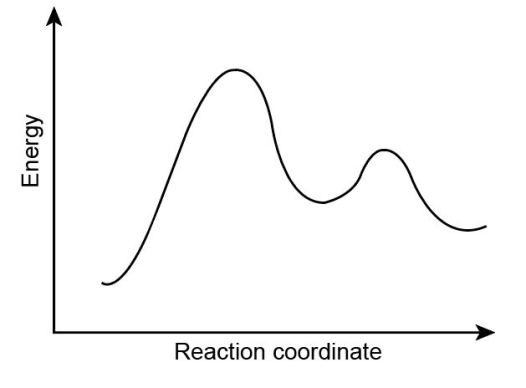
Consider the following energy diagram
The following is a concerted, bimolecular reaction:.
a. What is the rate equation for this reaction?
b. What happens to the rate of the reaction if is doubled?
c. What happens to the rate of the reaction if [NaCN] is halved?
d. What happens to the rate of the reaction if and [NaCN] are both increased by a factor of five?
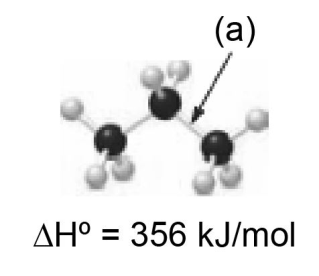
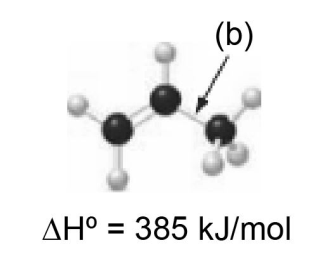
Explain why the bond dissociation energy for bond (a) is lower than the bond dissociation energy for bond (b).
What do you think about this solution?
We value your feedback to improve our textbook solutions.