Chapter 6: Problem 6.17 (page 231)
Draw the structure for the transition state in each reaction.
a.
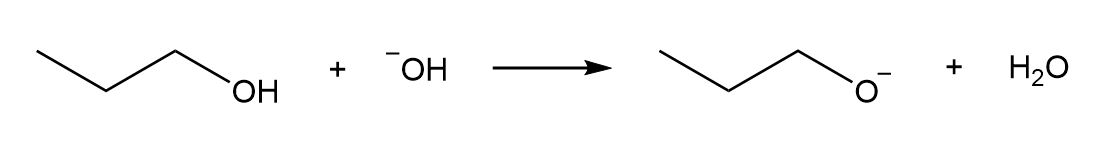
b.
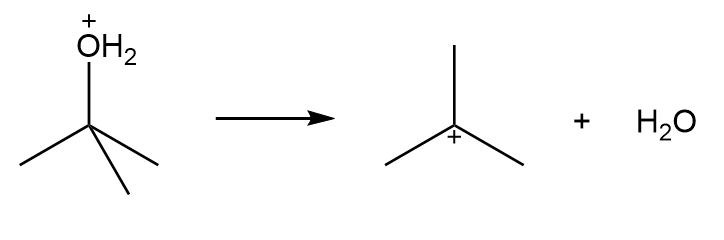
Short Answer
Answer
a.
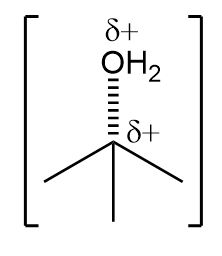
b.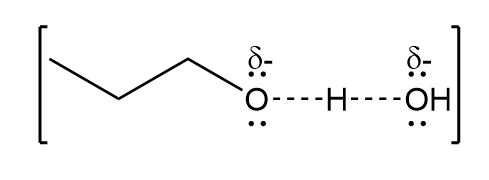
Learning Materials
Features
Discover
Chapter 6: Problem 6.17 (page 231)
Draw the structure for the transition state in each reaction.
a.

b.

Answer
a.

b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw an energy diagram for a reaction in which the products are higher in energy than the starting materials and is large. Clearly label all of the following on the diagram: the axes, the starting materials, the products, the transition state, ,and .
Classify each transformation as substitution, elimination, or addition.
a.
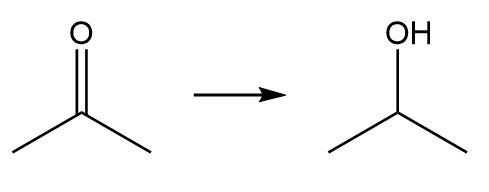
b.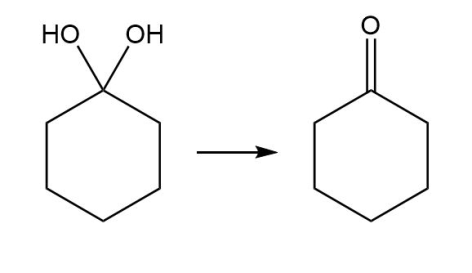
Classify each transformation as substitution, elimination, or addition.
a.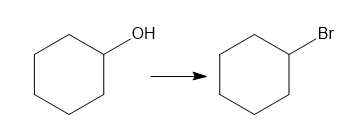
b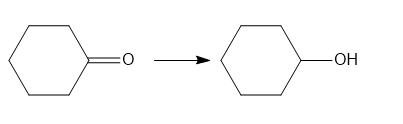
c.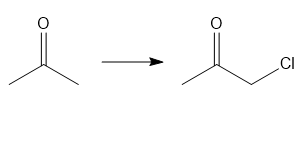
d.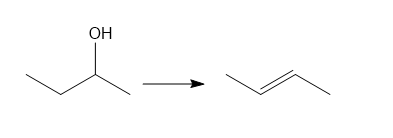
As we learned in Chapter 4, monosubstituted cyclohexanes exist as an equilibrium mixture of two conformations having either an axial or equatorial substituent. When , for this process is 23. When ,for this process is 4000.
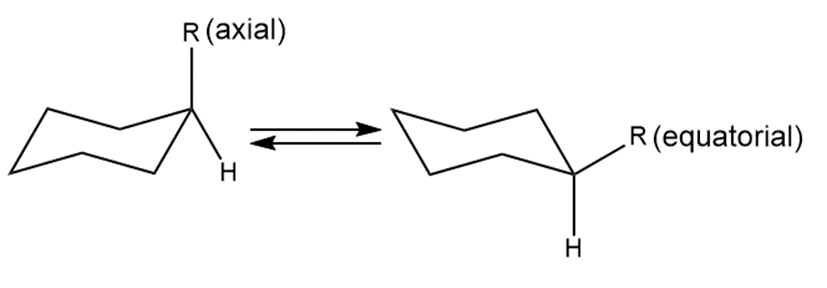
a. When , which conformation is present in higher concentration?
b. Which R shows the higher percentage of equatorial conformation at equilibrium?
c. Which R shows the higher percentage of axial conformation at equilibrium?
d. For which R is more negative?
e. How is the size of R related to the amount of axial and equatorial conformations at equilibrium?
Indicate which factors affect the rate of a reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.