Chapter 6: Problem 6.1 (page 216)
Classify each transformation as substitution, elimination, or addition.
a.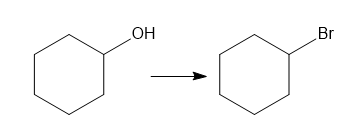
b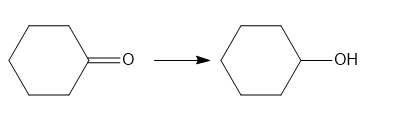
c.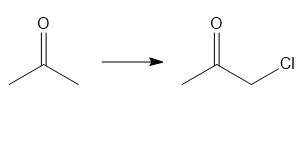
d.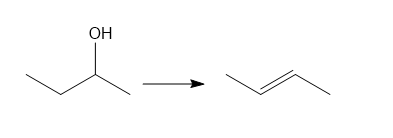
Short Answer
Answer
a. Substitution reaction.
b. Addition reaction.
c. Substitution reaction.
d. Elimination reaction.
Learning Materials
Features
Discover
Chapter 6: Problem 6.1 (page 216)
Classify each transformation as substitution, elimination, or addition.
a.
b
c.
d.
Answer
a. Substitution reaction.
b. Addition reaction.
c. Substitution reaction.
d. Elimination reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why is more acidic than , even though the C-H bond in has a higher bond dissociation energy than the C-H bond in .
Given each of the following values, is the starting material or product favored at equilibrium?
a.
b.
As we learned in Chapter 4, propane has both and hydrogens.
Draw an energy diagram for each reaction. Label the axes, the starting material, product, transition state, , and .
a.a concertedwith and .
b. a two-step reaction, , in which the relative energy of the compounds is , and the step is rate-determining.
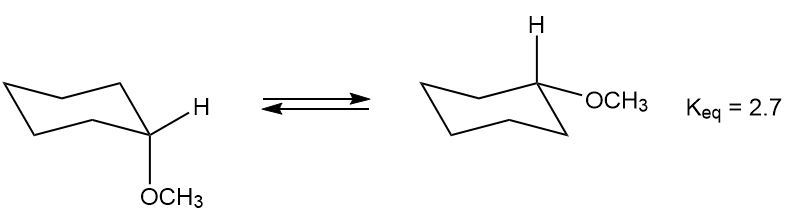
The equilibrium constant for the conversion of the axial to the equatorial conformation of methoxycyclohexane is 2.7.
What do you think about this solution?
We value your feedback to improve our textbook solutions.