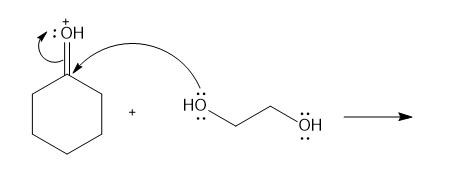
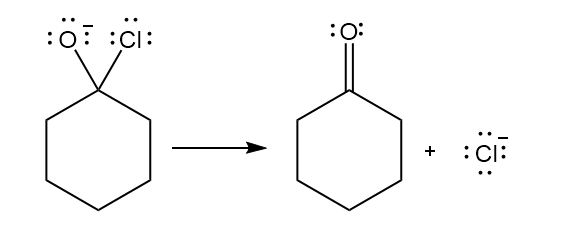
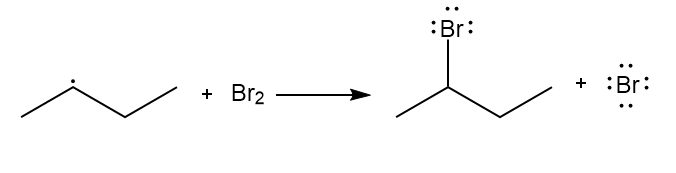
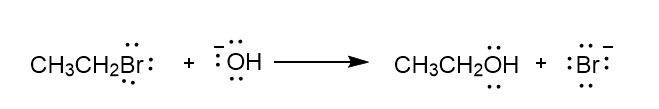
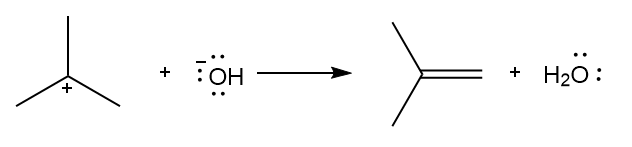
Chapter 6: Problem 6.13 (page 228)
Question: Considering each of the following values and neglecting entropy, tell whether the starting material or product is favored at equilibrium:
(a)
(b)
Short Answer
Answer
(a) Starting material is favored at equilibrium.
(b) Products are favored at equilibrium.