Chapter 6: Problem 6.15 (page 228)
Answer Problem 6.14 for a reaction with .
Short Answer
Answer
- True.
- False; for the reaction is negative.
- True.
- False; The bonds in the starting materials are weaker than the bonds in the product.
- True.
Learning Materials
Features
Discover
Chapter 6: Problem 6.15 (page 228)
Answer Problem 6.14 for a reaction with .
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider the following energy diagram for the conversion of .
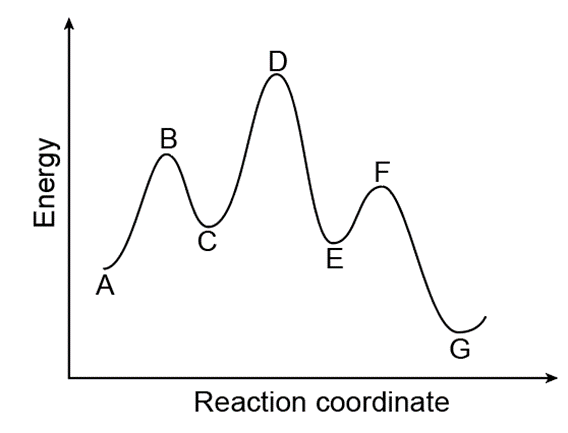
a. Which points on the graph correspond to transition states?
b. Which points on the graph correspond to reactive intermediates?
c. How many steps are present in the reaction mechanism?
d. Label each step of the mechanism as endothermic or exothermic.
e. Label the overall reaction as endothermic or exothermic.
The following is a concerted, bimolecular reaction:.
a. What is the rate equation for this reaction?
b. What happens to the rate of the reaction if is doubled?
c. What happens to the rate of the reaction if [NaCN] is halved?
d. What happens to the rate of the reaction if and [NaCN] are both increased by a factor of five?
For a reaction with , decide which of the following statements is (are) true. Correct any false statement to make it true. (a) The reaction is exothermic; (b) for the reaction is positive; (c) is greater than 1; (d) the bonds in the starting materials are stronger than the bonds in the product; and (e) the product is favored at equilibrium.
Calculate Δ��° for each reaction.
The Diels–Alder reaction, a powerful reaction discussed in Chapter 16, occurs when a 1,3- diene such as A reacts with an alkene such as B to form the six-membered ring in C.
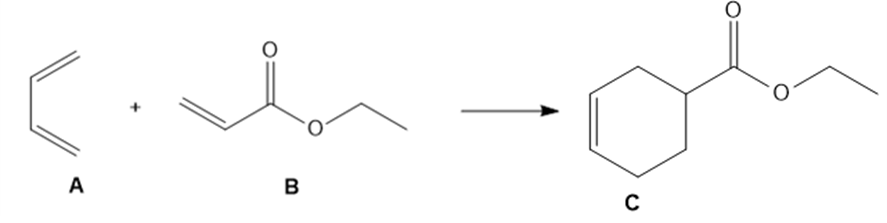
a. Draw curved arrows to show how A and B react to form C.
b. What bonds are broken and formed in this reaction?
c. Would you expect this reaction to be endothermic or exothermic?
d. Does entropy favor the reactants or products?
e. Is the Diels–Alder reaction a substitution, elimination, or addition?
What do you think about this solution?
We value your feedback to improve our textbook solutions.