Chapter 6: Problem 6.45 (page 243)
Consider the following energy diagram for the conversion of .
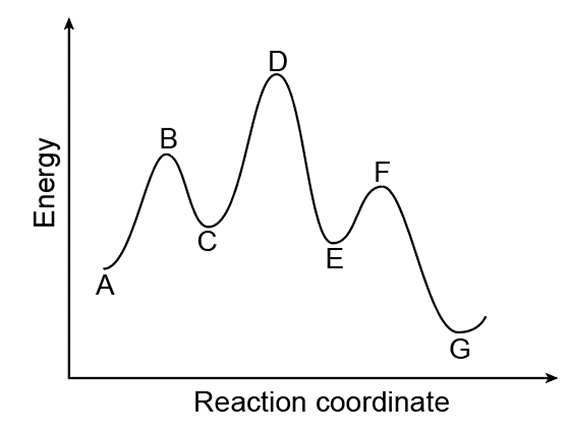
a. Which points on the graph correspond to transition states?
b. Which points on the graph correspond to reactive intermediates?
c. How many steps are present in the reaction mechanism?
d. Label each step of the mechanism as endothermic or exothermic.
e. Label the overall reaction as endothermic or exothermic.
Short Answer
Answer
- Points B, D, and F are the transition states.
- Points C and E are the reactive intermediates.
- The reaction has three steps.
- Step A-C is endothermic. While the steps C-E and E-G are exothermic.
- The overall reaction is exothermic.