Chapter 5: Q.38 (page 206)
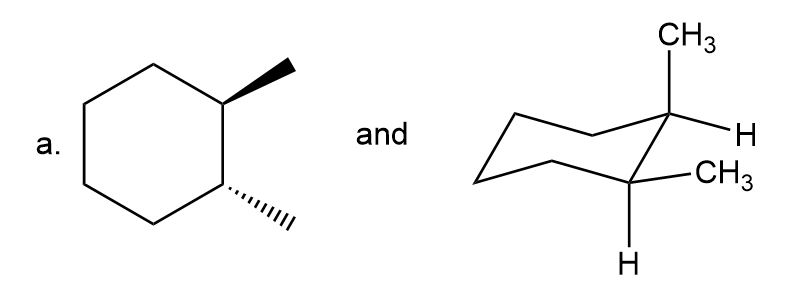
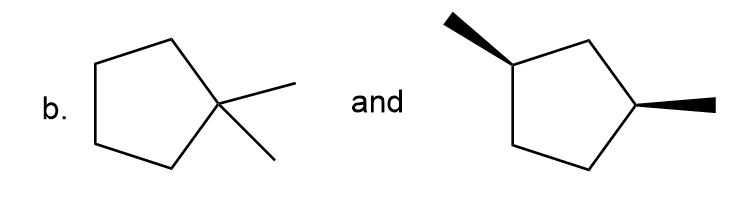
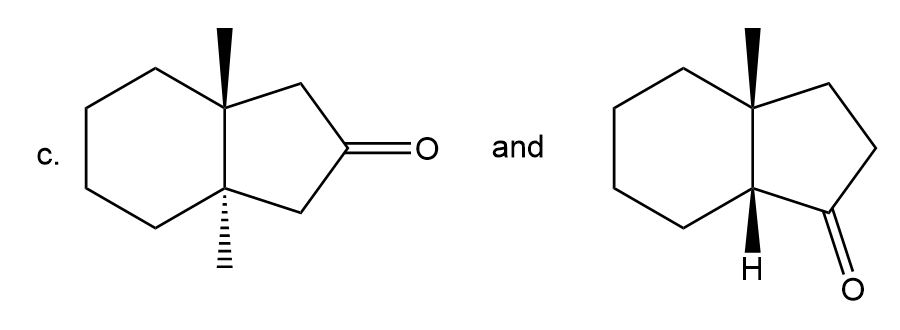
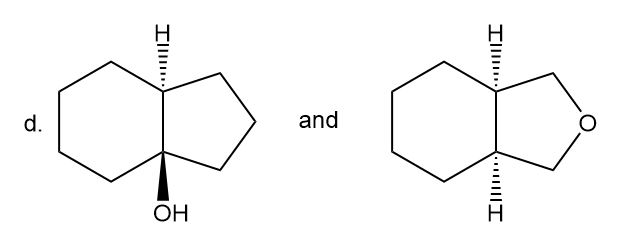
Label each pair of compounds as constitutional isomers, stereoisomers, or not isomers of each other.
Short Answer
- Stereoisomers
- Constitutional isomers
- Not isomers of each other
Not isomers of each other
Learning Materials
Features
Discover
Chapter 5: Q.38 (page 206)
Label each pair of compounds as constitutional isomers, stereoisomers, or not isomers of each other.




Not isomers of each other
All the tools & learning materials you need for study success - in one app.
Get started for free
If the two stereogenic centers of a compound are R,S in configuration, what are the R,S assignments for its enantiomer and two diastereomers?
Which group in each pair is assigned the higher priority in R,S nomenclature?
Draw all the possible stereoisomers for each compound and label pairs of enantiomers and diastereomers.
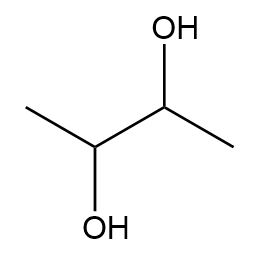
a.
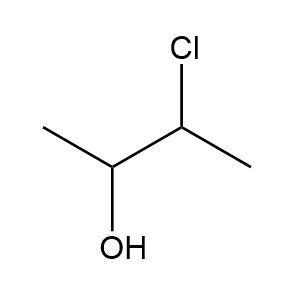
b.
Rank the following groups in order of decreasing priority.
Locate the stereogenic centers in each Newman projection and label each center as R or S.
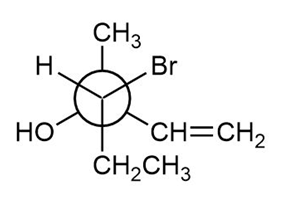
a.
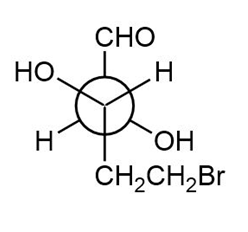
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.