Chapter 5: Q.19 (page 194)
Draw all the possible stereoisomers for each compound and label pairs of enantiomers and diastereomers.
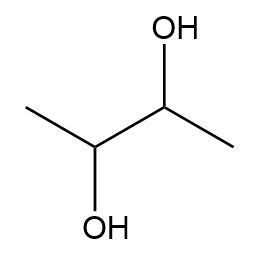
a.
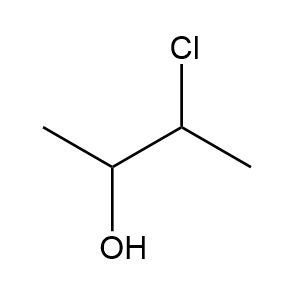
b.
Short Answer
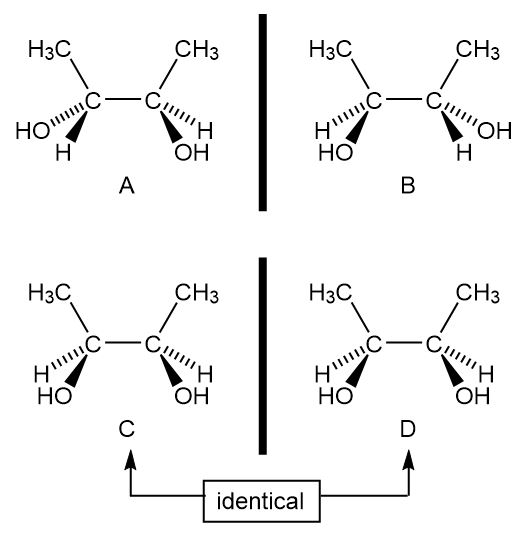
a.
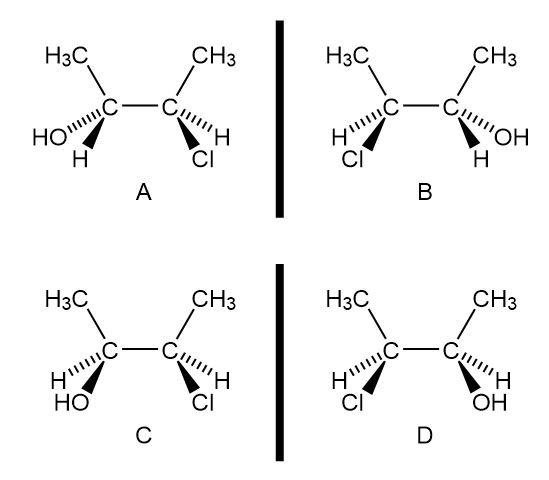
b.
Learning Materials
Features
Discover
Chapter 5: Q.19 (page 194)
Draw all the possible stereoisomers for each compound and label pairs of enantiomers and diastereomers.

a.

b.
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
An acid–base reaction of (R)-sec-butylamine with a racemic mixture of 2-phenylpropanoic acid forms two products having different melting points and somewhat different solubilities. Draw the structure of these two products. Assign R and S to any stereogenic centers in the products. How are the two products related? Choose from enantiomers, diastereomers, constitutional isomers, or not isomers.
Drawn are four isomeric dimethylcyclopropanes.
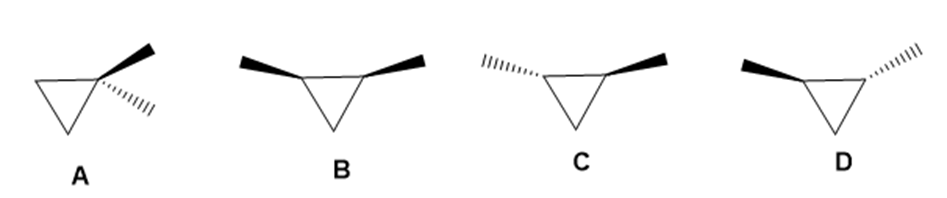
a. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?
b. Label each compound as chiral or achiral.
c. Which compounds, alone, would be optically active?
d. Which compounds have a plane of symmetry?
e. How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D?
f. Which of the compounds are meso compounds?
g. Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?
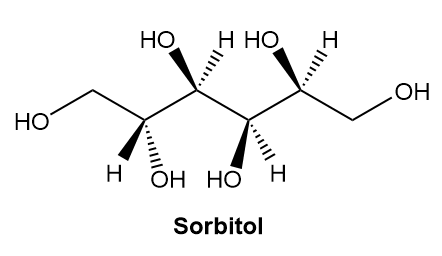
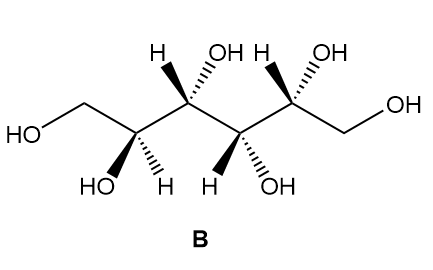
a. Label the four stereogenic centres in sorbitol as R or S.
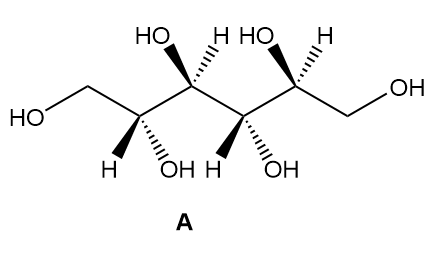
b. How are sorbitol and A related?
c. How are sorbitol and B related?
Artemisinin and mefloquine are widely used antimalarial drugs. A ball-and-stick model of artemisinin appears on the cover of this text.
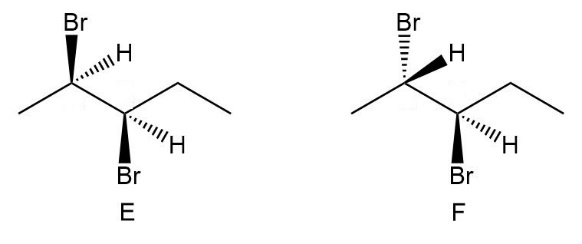
Compounds E and F are two isomers of 2,3-dibromopentane drawn in staggered conformations. Which compounds (A–D) in Figure 5.8 are identical to E and F?
What do you think about this solution?
We value your feedback to improve our textbook solutions.