Chapter 15: Q61. (page 570)
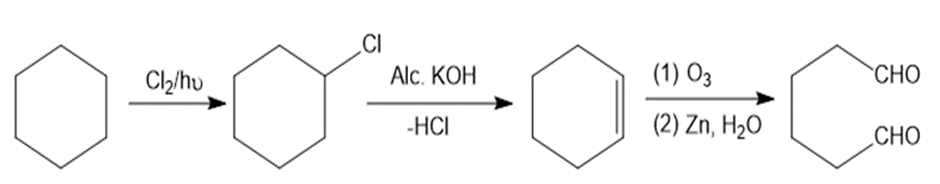
Question. Devise a synthesis of OHC(CH2)4CHO from cyclohexane using any required organic or inorganic reagents.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 15: Q61. (page 570)
Question. Devise a synthesis of OHC(CH2)4CHO from cyclohexane using any required organic or inorganic reagents.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
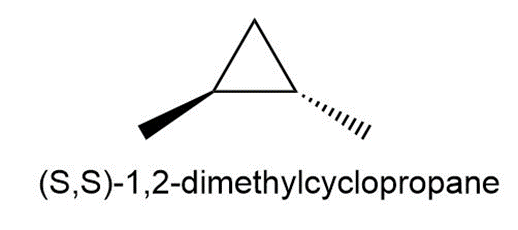
Question:Draw all the monochlorination products that are formed from (S,S)-1,2-dimethylcyclopropane.
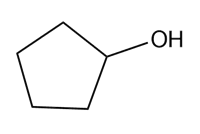
Question: Devise a synthesis of each compound from cyclopentane and any other required organic or inorganic reagents.
a.
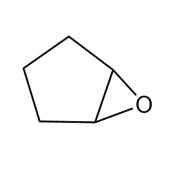
b.
c.
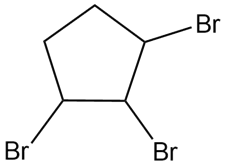
d.
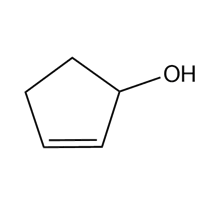
e.
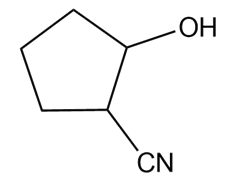
Question: Although CH4 reacts with Cl2 to form CH3Cl and HCl, the corresponding reaction of CH4with l2does not occur at an appreciable rate, even though the bond is much weaker than the bond. Explain why this is so.
Question: Draw the structure of the four allylic halides formed when 3-methylcyclohexene undergoes allylic halogenation with NBS + hν.
Question: With reference to the indicated C-H bonds in 2-methylbutane.
a. Rank the C-H bonds in order of increasing bond length.
b. Draw the radical resulting from cleavage of each C-H bond and classify it as 10,20, or 30
c.Rank the order of radicals in order of increasing stability.
Rank the C-H bonds in order of increasing ease of H abstractions in a radical halogenation reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.