Chapter 15: Q62. (page 570)
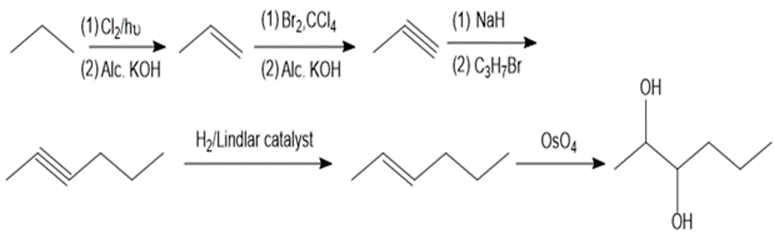
Question. Devise a synthesis of hexane-2,3-diol from propane as the only source of carbon atoms. You may use any other required organic or inorganic reagents.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 15: Q62. (page 570)
Question. Devise a synthesis of hexane-2,3-diol from propane as the only source of carbon atoms. You may use any other required organic or inorganic reagents.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
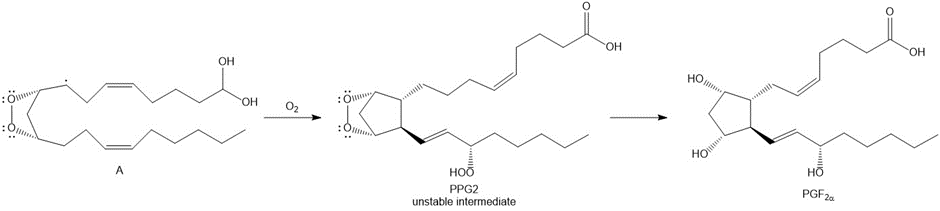
Question: PGF2 (Section 4.15) is synthesized in cells from arachidonic acid C20H32O2 using a cyclooxygenase enzyme that catalyzes a multistep radical pathway. Part of this process involves the conversion of radical A to PGG2, an unstable intermediate, which is then transformed to PGF2 and other prostaglandins. Draw a stepwise mechanism for the conversion of A to PGG2 . (Hint: The mechanism begins with radical addition to a carbon-carbon double bond to form a resonance-stabilized radical.
Question: Nitric oxide, NO•, is another radical also thought to cause ozone destruction by a similar mechanism. One source of NO• in the stratosphere is supersonic aircraft whose jet engines convert small amounts of N2 and O2 to NO•. Write the propagation steps for the reaction of O3 with NO•
Question: Draw the products formed when a chlorine atom reacts with each species.
a.
b.
c.
d. O2
Question: Although CH4 reacts with Cl2 to form CH3Cl and HCl, the corresponding reaction of CH4with l2does not occur at an appreciable rate, even though the bond is much weaker than the bond. Explain why this is so.
Question: Devise a synthesis of 1-methylcyclohexene oxide from methylcyclohexane. You may use any other required organic or inorganic reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.