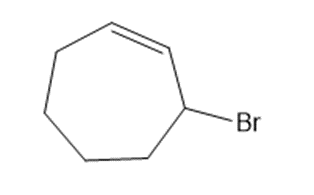
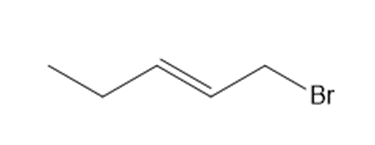
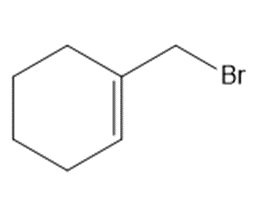
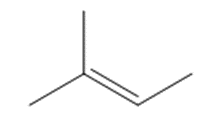
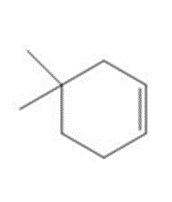
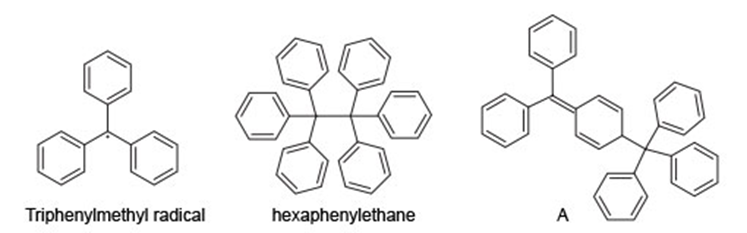
Chapter 15: Q15. (page 570)
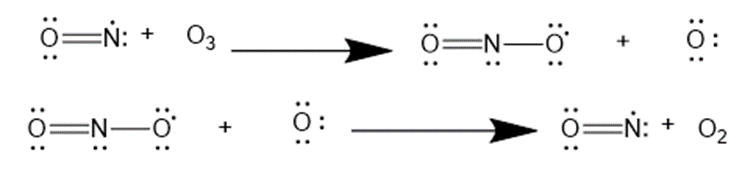
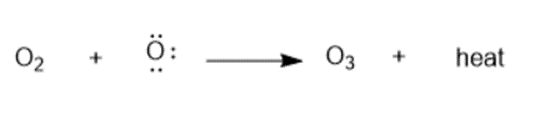
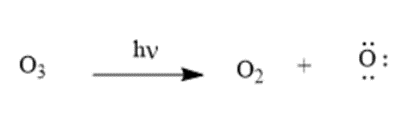
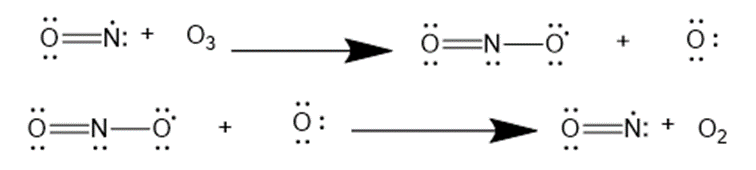
Question: Nitric oxide, NO•, is another radical also thought to cause ozone destruction by a similar mechanism. One source of NO• in the stratosphere is supersonic aircraft whose jet engines convert small amounts of N2 and O2 to NO•. Write the propagation steps for the reaction of O3 with NO•
Short Answer
Answer