Chapter 15: Q12. (page 570)
Question: Show all steps and reagents needed to convert cyclohexane into each compound: (a) the two enantiomers of trans-1,2-dibromocyclohexane; and (b) 1,2-epoxycyclohexane.
Short Answer
Answer
(a)
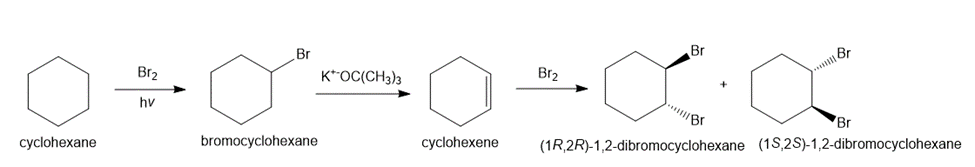
(b)
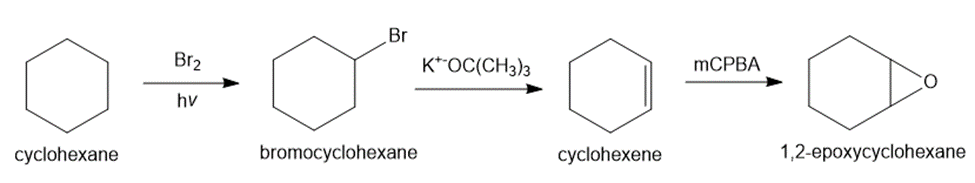
Learning Materials
Features
Discover
Chapter 15: Q12. (page 570)
Question: Show all steps and reagents needed to convert cyclohexane into each compound: (a) the two enantiomers of trans-1,2-dibromocyclohexane; and (b) 1,2-epoxycyclohexane.
Answer
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw a second resonance structure for each radical. Then draw the hybrid.
a.
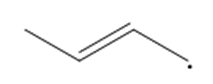
b.
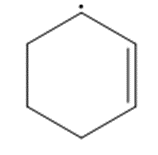
c.
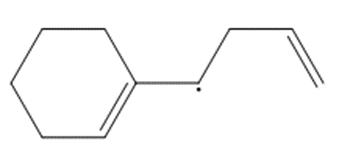
d.
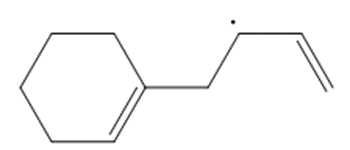
Question: Nitric oxide, NO•, is another radical also thought to cause ozone destruction by a similar mechanism. One source of NO• in the stratosphere is supersonic aircraft whose jet engines convert small amounts of N2 and O2 to NO•. Write the propagation steps for the reaction of O3 with NO•
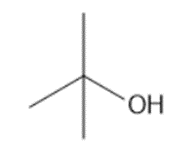
Question: Synthesize each compound from (CH3)3 CH.
a.
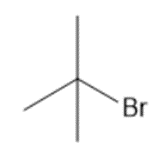
b.
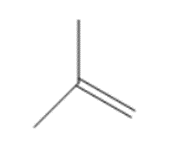
c.
Question: Calculate H0 for the rate-determining step of the reaction of CH4 with I2. Explain why this result illustrates that this reaction is extremely slow.
Question: Draw all constitutional isomers formed by monochlorination of each alkane with Cl2 and h.
What do you think about this solution?
We value your feedback to improve our textbook solutions.